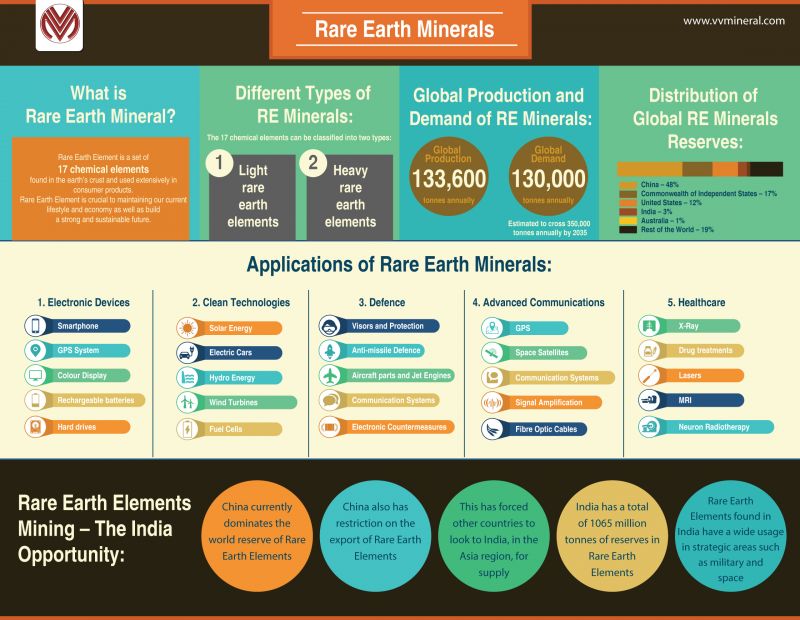
- The rare earth elements (REE) are a set of seventeen metallic elements. These include the fifteen lanthanides on the periodic table in addition to scandium and yttrium that show similar physical and chemical properties to the lanthanides
- The REEs have unique catalytic, metallurgical, nuclear, electrical, magnetic and luminescent properties.
- While named ‘rare earth’, they are in fact not that rare and are relatively abundant in the Earth’s crust
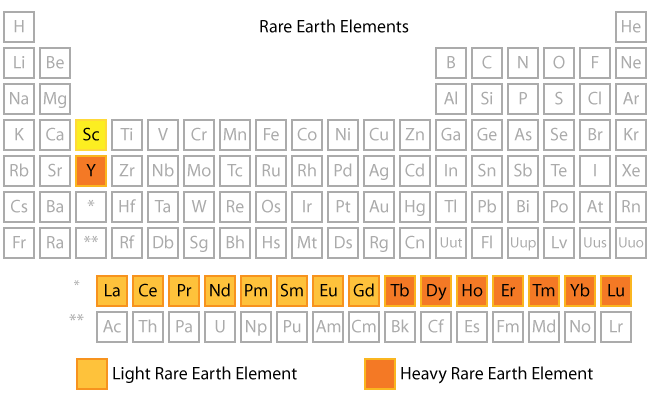
Common Properties of REE
- The rare earths are silver, silvery-white, or gray metals.
- The metals have a high luster, but tarnish readily in air.
- The metals have high electrical conductivity.
- The rare earths share many common properties. This makes them difficult to separate or even distinguish from each other.
- There are very small differences in solubility and complex formation between the rare earths.
- The rare earth metals naturally occur together in minerals (e.g., monazite is a mixed rare earth phosphate).
Distribution of REE

- China currently accounts for an overwhelming 97% of global production.
- India currently has a little over 2% share of global output of rare earths.
- Kerala, Orissa and Tamil Nadu account for nearly 95% of the country’s production of rare earths.

Institutional Setup
Indian Rare Earths Limited (IREL) was incorporated on August 18, 1950 with its first unit Rare Earths Division (RED), Aluva in Kerala. It became a full-fledged Government of India Undertaking, under the administrative control of Department of Atomic Energy (DAE) in year 1963 and took over companies engaged in mining and separation of beach sand minerals in southern part of the country, by establishing two more divisions one at Chavara, Kerala and the other at Manavalakurichi (MK),Tamilnadu.
