What are IPRs?
Intellectual Property Rights (IPRs) are legal rights, which result from intellectual invention, innovation and discovery in the industrial, scientific, literary and artistic fields. These rights entitle an individual or group to the moral and economic rights of creators in their creation.
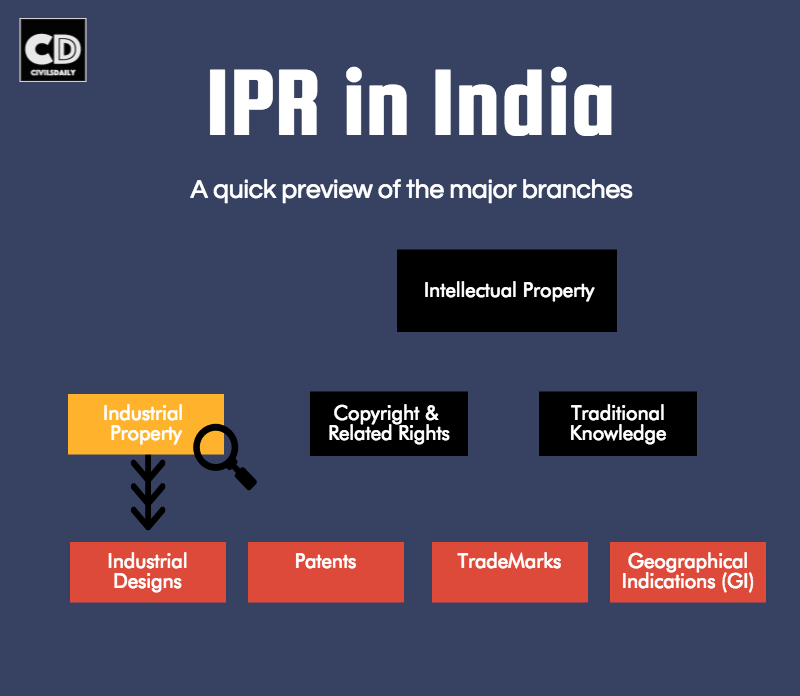
Types:
Patent- It is a set of exclusive rights granted by a sovereign state to an inventor for a limited period of time in exchange for detailed public disclosure of an invention.
Copyright- It is a legal right created by the law of a country that grants the creator of an original work exclusive rights for its use and distribution. It includes literary & artistic works such as novels, poems, plays, films, musical works, drawing, painting, photography, sculpture, architectural designs
Trademark- It is a recognizable sign, design, or expression which identifies products or services of a particular source from those of others. Trademarks used to identify services are usually called service marks.
Industrial design right- It is an intellectual property right that protects the visual design of objects that are not purely utilitarian. An industrial design consists of the creation of a shape, configuration or composition of pattern or color, or combination of pattern and color in three-dimensional form containing aesthetic value. An industrial design can be a two- or three-dimensional pattern used to produce a product, industrial commodity or handicraft.
Trade secret- It is a formula, practice, process, design, instrument, pattern, commercial method, or compilation of information which is not generally known or reasonably ascertainable by others, and by which a business can obtain an economic advantage over competitors or customers
Geographical Indication (GI)- It is a name or sign used on certain products which corresponds to a specific geographical location or origin (e.g. a town, region, or country). The use of a geographical indication may act as a certification that the product possesses certain qualities, is made according to traditional methods, or enjoys a certain reputation, due to its geographical origin. A recent example is of Indian variety of Basmati rice getting GI tag.
From above points, it is clear that IPR is a very sensitive issue in terms of businesses different kinds and international relations as well.
IPRs in pharmaceutical sector:
Some sectors are very sensitive in terms of IPRs like pharmaceuticals. Let’s explore briefly into IPR issues in pharmaceutical sector.
We hear of two kinds of drugs- generic and brand name drugs:
Generic drugs are those whose patent has expired or does not exist and which can be produced by any registered manufacturer without need of taking permission from any authority and also without any payment of royalty.
Brand name drugs are those which are patented and cannot be produced without the consent of the patent holder. A royalty is to be paid for production of these drugs.
But what happens if a company holds patent of an essential drug and there is an emergency in which the drug needs to be provided at low cost for vast populace? In this case, Compulsory Licensing comes to the rescue.
What is Compulsory Licensing?
- A compulsory license provides that the owner of a patent or copyright licenses the use of their rights against a payment. This payment is either set by law or determined through some form of arbitration
- In essence, under a compulsory license, an individual or company seeking to use another’s intellectual property can do so without seeking the rights holder’s consent, and pays the rights holder a set fee for the license
- This is an exception to the general rule under intellectual property laws that the intellectual property owner enjoys exclusive rights that it may license – or decline to license – to others
Does there have to be an emergency?
Not necessarily. This is a common misunderstanding. The TRIPS Agreement does not specifically list the reasons that might be used to justify compulsory licensing. However, the Doha Declaration on TRIPS and Public Health confirms that countries are free to determine the grounds for granting compulsory licences.
In March 2012, India granted its first compulsory license ever. The license was granted to Indian generic drug manufacturer Natco Pharma Ltd for Sorafenib tosylate, a cancer drug patented by Bayer.
Here, first thing first, What is TRIPS?
- TRIPS is an international agreement administered by the World Trade Organization (WTO), which sets down minimum standards for many forms of intellectual property (IP) regulations as applied to the nationals of other WTO Members
- It was negotiated at the end of the Uruguay Round of the General Agreement on Tariffs and Trade (GATT) in 1994
- TRIPS requires WTO members to provide copyright rights, covering content producers including performers, producers of sound recordings and broadcasting organizations, geographical indications, including appellations of origin, industrial designs, integrated circuit layout-designs, patents, new plant varieties, trademarks, trade dress, and undisclosed or confidential information
- The agreement also specifies enforcement procedures, remedies, and dispute resolution procedures
Now, back to the topic…
India is a huge market for generic drugs and hence it is very obvious that there must emerge issues out of patents for pharmaceuticals.
One such case came up in 1998- Novartis v. Union of India & Others
It was a landmark decision by a two-judge bench of the Supreme Court, on the issue of whether Novartis could patent Glivec in India. It was the culmination of a seven-year-long litigation fought by Novartis. The Supreme Court upheld the Indian patent office’s rejection of the patent application.
Ground of rejection?
Novartis claimed patent for he changed form of Glivec on the basis of the increased bio-availability in the body of the patient by making changes in chemical composition of its original anti-cancer drug Imatinib Mesylate. This changed form of the drug could not withstand the ‘enhanced therapeutic efficacy’ test enshrined under Section 3(d) of Indian Patents Act and therefore it was rejected.
Recently, Gilead got patent for its Hepatitis C drug Solvadi. An application for the same patent was first rejected in January 2015 as lacking inventiveness and novelty. The decision, however, is seen as a major blow to the access to drug movement
Now let’s turn towards the latest developments in the IPRs in India.
New IPR Policy
Govt of India recently released a new National Intellectual Property Rights (IPR) Policy which is in compliance with WTO’s agreement on TRIPS
Why a new policy?
- Global drug brands led by US companies have been pushing for changes to India’s intellectual property rules for quite some time now. They have often complained about India’s price controls and marketing restrictions
- Also, an IPR policy is important for the government to formulate incentives in the form of tax concessions to encourage research and development (R&D)
- It is also critical to strengthen the Make In India, Startup and Digital India schemes
- The IPR policy comes at a time when India and other emerging countries faces fresh challenges from the developed world and mega regional trade agreements such as the Trans-Pacific Partnership (TPP)
Seven objectives:
- IPR Awareness: To create public awareness about the economic, social and cultural benefits of IPRs among all sections of society
- Generation of IPRs: To stimulate the generation of IPRs
- Legal and Legislative Framework: To have strong and effective IPR laws, which balance the interests of rights owners with larger public interest
- Administration and Management: To modernize and strengthen service-oriented IPR administration
- Commercialization of IPRs: Get value for IPRs through commercialization
- Enforcement and Adjudication: To strengthen the enforcement and adjudicatory mechanisms for combating IPR infringements
- Human Capital Development: To strengthen and expand human resources, institutions and capacities for teaching, training, research and skill building in IPRs
Highlights:
- The new policy calls for providing financial support to the less empowered groups of IP owners or creators such as farmers, weavers and artisans through financial institutions like rural banks or co-operative banks offering IP-friendly loans
- The work done by various ministries and departments will be monitored by the Department of Industrial Policy & Promotion (DIPP), which will be the nodal department to coordinate, guide and oversee implementation and future development of IPRs in India
- The policy, with a tagline of Creative India: Innovative India, also calls for updating various intellectual property laws, including the Indian Cinematography Act, to remove anomalies and inconsistencies in consultation with stakeholders
- For supporting financial aspects of IPR commercialisation, it asks for financial support to develop IP assets through links with financial institutions, including banks, VC funds, angel funds and crowd-funding mechanisms
- To achieve the objective of strengthening enforcement and adjudicatory mechanisms to combat IPR infringements, it called for taking actions against attempts to treat generic drugs as spurious or counterfeit and undertake stringent measures to curb manufacture and sale of misbranded, adulterated and spurious drugs
- The policy will be reviewed after every five years to keep pace with further developments in the sector
International angle:
Last month, the US Trade Representative kept India, China and Russia on its “Priority Watch List” for inadequate improvement in IPR protection. However, brushing aside concerns of the US on India’s IPR regime, the government said its intellectual property rights laws are legal-equitable and WTO-compliant. Thus, the government has not yielded to pressure from the United States to amend India’s patent laws.
Benefits:
- The new policy will try to safeguard the interests of rights owners with the wider public interest, while combating infringements of intellectual property rights
- By 2017, the window for trademark registration will be brought down to one month. This will help in clearing over 237,000 pending applications in India’s four patent offices
- It also seeks to promote R&D through tax benefits available under various laws and simplification of procedures for availing of direct and indirect tax benefits
- Unlike earlier where copyright was accorded to only books and publications, the recast regime will cover films, music and industrial drawings
- A host of laws will also be streamlined — on semi-conductors, designs, geographical indications, trademarks and patents
- The policy also puts a premium on enhancing access to healthcare, food security and environmental protection
- Policy will provide both domestic and foreign investors a stable IPR framework in the country
- This will promote a holistic and conducive ecosystem to catalyse the full potential of intellectual property for India’s growth and socio-cultural development while protecting public interest
- It is expected to lay the future roadmap for intellectual property in India, besides putting in place an institutional mechanism for implementation, monitoring and review
- The idea is to incorporate global best practices in the Indian context and adapt to the same
Challenges:
- According to the policy, India will retain the right to issue so-called compulsory licenses to its drug firms, under “emergency” conditions
- Also, the government has indicated that there is no urgent need to change patent laws that are already fully World Trade Organization-compliant. So India has resisted pressure from the US and other Western countries to amend its patent laws
- The policy also specifically does not open up Section 3(d) of the Patents Act, which sets the standard for what is considered an invention in India, for reinterpretation
Published with inputs from Swapnil