Why in the News?
The President of India has approved the conferment of the Jeevan Raksha Padak Series of Awards 2025 to 30 individuals for acts of exceptional courage in saving lives.
- 6 Sarvottam Jeevan Raksha Padak
- 6 Uttam Jeevan Raksha Padak
- 18 Jeevan Raksha Padak
- 6 awards are posthumous
What are the Jeevan Raksha Padak Awards?
- A civilian life saving gallantry award series
- Recognises meritorious acts of humane nature involving personal risk to save another person’s life
- Established in 1961
- Originated as an offshoot of the Ashoka Chakra series of Gallantry Awards
Categories
- Sarvottam Jeevan Raksha Padak: Conspicuous courage in saving life under very great danger to the rescuer
- Uttam Jeevan Raksha Padak: Courage and promptitude under great danger to the rescuer
- Jeevan Raksha Padak: Courage and promptitude involving grave risk of bodily injury to the rescuer
Eligibility
- Open to persons of all genders and walks of life
- Posthumous awards permitted
- Acts considered include rescues during Drowning, Fires, Accidents, Electrocution, Mine rescues and Natural calamities.
| [2021] Consider the following statements in respect of Bharat Ratna and Padma Awards:
1. Bharat Ratna and Padma Awards are titles under Article 18(1) of the Constitution of India.
2. Padma Awards, which were instituted in the year 1954, were suspended only once
3. The number of Bharat Ratna Awards is restricted to a maximum of five in a particular year.
Which of the above statements are not correct?
(a) 1 and 2 only (b) 2 and 3 only (c) 1 and 3 only (d) 1, 2, and 3 |
Why in the News?
The Padma Awards 2026 were announced on the eve of Republic Day, with the President of India approving 131 awards across the Padma Vibhushan, Padma Bhushan, and Padma Shri categories.
What are the Padma Awards?
- Among India’s highest civilian honours
- Conferred for distinguished and exceptional service with a clear element of public service
- Instituted in 1954 by the Government of India
Historical Background
- Initially, two civilian awards were instituted in 1954
- Bharat Ratna
- Padma Vibhushan
- In 1955, Padma Vibhushan was reclassified into three categories
- Padma Vibhushan
- Padma Bhushan
- Padma Shri
- Awards are announced annually on Republic Day
- Not conferred during 1978 to 1979 and 1993 to 1997
Categories and Purpose
- Padma Vibhushan: Exceptional and distinguished service
- Padma Bhushan: Distinguished service of a high order
- Padma Shri: Distinguished service in any field
Eligibility Criteria
- Open to all persons, irrespective of race, gender, occupation, or position
- Government servants, including PSU employees, are generally not eligible
- Exception for doctors and scientists
- Normally not awarded posthumously
- Permitted in exceptional cases
- Minimum gap of 5 years required for a higher Padma category
- Can be relaxed in deserving cases
- Award recognises excellence plus, not merely long service
Fields Recognised
- Art, Social Work, Public Affairs, Science and Engineering, Trade and Industry, Medicine, including AYUSH, Literature and Education, Civil Service, Sports and Others, such as culture, environment, wildlife conservation, and human rights
| Consider the following statements in respect of Bharat Ratna and Padma Awards:
1. Bharat Ratna and Padma Awards are titles under Article 18(1) of the Constitution of India.
2. Padma Awards, which were instituted in the year 1954, were suspended only once
3. The number of Bharat Ratna Awards is restricted to a maximum of five in a particular year.
Which of the above statements are not correct?
(a) 1 and 2 only (b) 2 and 3 only (c) 1 and 3 only (d) 1, 2, and 3 |
Introduction
The 2025 Sveriges Riksbank Prize in Economic Sciences was awarded to Joel Mokyr, Philippe Aghion, and Peter Howitt for “explaining innovation-driven economic growth.” Their research collectively answers one of the most fundamental economic puzzles — how nations sustain growth over centuries, not decades.
Why in the News
The Nobel Committee’s decision is significant because it celebrates innovation as the engine of sustained prosperity at a time when economies face stagnation despite technological abundance. It also marks a historical synthesis, combining Mokyr’s economic history with Aghion and Howitt’s modern growth models, to offer a unified vision of why the last two centuries broke free from millennia of stagnation. This award underscores that knowledge creation and openness to change are as critical to a nation’s future as natural resources or fiscal policy.
Understanding the Foundations of Innovation-Driven Growth
What did Joel Mokyr’s research reveal about sustained growth?
- Useful Knowledge: Mokyr argued that long-term growth depends on a constant flow of useful knowledge, divided into propositional (theoretical understanding) and prescriptive (practical implementation) forms.
- Before Industrial Revolution: Innovators understood why things worked (propositional) but lacked the technical ability to make them work (prescriptive).
- Scientific Revolution Impact: The 16th–17th centuries brought controlled experiments and reproducibility — transforming knowledge from abstract to applicable.
- Policy Implication: Nations must ensure technical education and skill development, as ideas alone cannot yield growth without implementation.
How did Mokyr link innovation to social openness?
- Openness to Change: Innovation often disrupts existing systems and creates losers; societies resistant to change stifle progress.
- Historical Example: Britain’s sustained growth stemmed from skilled artisans and engineers who translated scientific ideas into industrial applications.
- Policy Lesson: Governments must create inclusive ecosystems that accept change, retrain workers, and redistribute gains from innovation.
What is the Theory of Creative Destruction?
- Conceptual Core: Originally introduced by Schumpeter, “creative destruction” describes how innovation replaces older technologies and firms, creating both winners and losers.
- Aghion & Howitt’s Contribution: They formalized this process mathematically, showing how technological progress leads to sustained long-term growth.
- Dynamic Equilibrium: Innovation raises productivity but simultaneously displaces outdated industries — a perpetual cycle that fuels development.
How much should a country invest in Research and Development (R&D)?
- Balancing Act: Aghion and Howitt’s model shows two opposing trends:
- Trend 1 — Underinvestment: Since society benefits from outdated technologies even after firms lose profits, R&D should be subsidized to ensure social spillovers.
- Trend 2 — Overinvestment: When incremental innovations capture disproportionate profits, R&D may be excessive and distort competition.
- Optimal Level: There is no universal ideal investment, but the model provides tools to identify an economy-specific optimum that maximizes welfare without creating monopolistic inefficiencies.
Why does this Nobel matter for developing economies like India?
- Knowledge Ecosystem: The laureates’ findings emphasise that growth requires not just innovation, but translation — turning ideas into scalable realities through skills, entrepreneurship, and openness.
- India’s Imperative: Investments in R&D (currently ~0.7% of GDP), vocational skilling, and ease of doing business are crucial to realize the demographic dividend.
- Policy Relevance: The Economic Survey and NITI Aayog’s “Innovation Index” already underline similar principles — this Nobel reinforces India’s need to build a “knowledge economy.”
Conclusion
The 2025 Nobel Prize in Economic Sciences reaffirms that innovation, knowledge, and societal openness are the real engines of prosperity. Economic success is no longer a product of mere capital or labor, but of the synergy between imagination and execution. For India and other developing nations, the message is clear: sustained growth depends on nurturing human capital, research ecosystems, and tolerance for disruption. As Mokyr’s and Aghion–Howitt’s work shows, societies that embrace change, skill their people, and invest in ideas will lead the next chapter of human progress.
PYQ Relevance
[UPSC 2015] What are the areas of prohibitive labour that can be sustainably managed by robots? Discuss the initiatives that can propel the research in premier research institutes for substantive and gainful innovation.
Linkage: This PYQ aligns with the 2025 Nobel Prize in Economic Sciences as both emphasize how technological innovation transforms labour structures—echoing Aghion and Howitt’s theory of creative destruction, where automation replaces old forms of work while driving new productivity.
Why in the News?
The 2025 Nobel Prize in Economic Sciences (Sveriges Riksbank Prize in Memory of Alfred Nobel) was awarded to Joel Mokyr (Northwestern University, US), Philippe Aghion (Collège de France, INSEAD, LSE), and Peter Howitt (Brown University, US) for their pioneering explanations of innovation-driven economic growth.
What is the Nobel Economics Prize?
- Officially called the Bank of Sweden Prize in Economic Sciences in Memory of Alfred Nobel, established in 1968.
- It is NOT part of the original Nobel Prizes created by Alfred Nobel in 1895.
- Created by the Swedish central bank to honor Alfred Nobel’s legacy.
- Although not an original Nobel Prize, it is presented alongside the other Nobel Prizes on December 10, the anniversary of Nobel’s death.
- Includes a diploma, gold medal, and a one-million-dollar prize for the laureates.
Who are the Nobel Laureates for 2025?
- Joel Mokyr (Northwestern University, USA): An economic historian, renowned for studying how scientific knowledge, cultural openness, and institutional change during the Enlightenment triggered the Industrial Revolution.
- Philippe Aghion (Collège de France, INSEAD, LSE): A leading growth theorist, known for advancing the Schumpeterian model of innovation-driven growth and the economics of creative destruction.
- Peter Howitt (Brown University, USA): Collaborator of Aghion, co-developer of the Aghion–Howitt growth model, integrating firm-level innovation dynamics into macroeconomic theory.
Their Contributions:
- Joel Mokyr:
- Demonstrated that before the 18th century, societies possessed “prescriptive knowledge” (how things worked) but lacked “propositional knowledge” (why they worked).
- Showed that the Scientific Revolution merged science with craftsmanship, turning discovery into applied innovation.
- Highlighted that the Enlightenment’s intellectual openness enabled acceptance of “creative destruction,” allowing new technologies to replace old ones without institutional backlash.
- Philippe Aghion & Peter Howitt:
- Developed the 1992 Schumpeterian Growth Model, mathematically linking innovation, competition, and economic growth.
- Explained that constant firm turnover—where new innovators replace old incumbents—creates long-term, stable growth.
- Introduced the idea of “general equilibrium in innovation”, connecting household savings, financial markets, R&D investment, and production into a single dynamic framework.
Why in the News?
Maria Corina Machado won the 2025 Nobel Peace Prize for defending democracy in Venezuela; President Trump praised her but criticised the Nobel Committee.

About Nobel Peace Prize:
- Origin: Instituted in 1901 under the will of Alfred Nobel, Swedish inventor and philanthropist, to honour outstanding contributions to peace and humanitarian cooperation.
- Administered By: Managed by the Norwegian Nobel Committee, a five-member body appointed by the Parliament of Norway, distinct from Sweden’s Nobel institutions.
- Purpose: Awards individuals or organisations advancing disarmament, peace negotiations, democracy, human rights, and a stable global order.
- Expanded Focus: Now includes climate change, environmental protection, and global justice as integral to sustainable peace.
- Prize Components: Laureates receive a gold medal, diploma, and 11 million Swedish krona (≈ US $1.2 million, 2025).
- Venue: Presented in Oslo, Norway, the only Nobel Prize awarded outside Sweden, symbolising Norway’s neutral and humanitarian tradition.
- Global Significance: Remains the world’s most prestigious peace honour, mirroring contemporary geopolitical and ethical realities.
These trivial facts are too unlikely to be asked in the CS prelims but may hold importance for CAPF and other exams.
US Presidents who won Nobel Peace Prize:
- Theodore Roosevelt (1906): Mediated the Russo–Japanese War settlement; first US President to win the prize.
- Woodrow Wilson (1919): Recognised for ending World War I and founding the League of Nations, precursor to the UN.
- Jimmy Carter (2002): Cited for human-rights mediation and the Camp David Accords, plus global work via the Carter Center.
- Al Gore (2007): Shared with the IPCC for elevating climate change as a global peace and security issue.
- Barack Obama (2009): Honoured for efforts toward nuclear disarmament and renewed international diplomacy; only US President got awarded while in office.
|
Why in the News?
The 2025 Nobel Prize in Literature has been awarded to Laszlo Krasznahorkai, a Hungarian novelist known for his dense, philosophical narratives and apocalyptic vision of modern existence.
Back2Basics: Nobel Prize in Literature
- First awarded in 1901, the Nobel Prize in Literature has been conferred 117 times to 121 laureates.
- Prize Details (2025): Each laureate receives 11 million Swedish kronor (~1.2 million USD), an 18-karat gold medal, and a diploma.
- Ceremony: Held annually on December 10, marking the death anniversary of Alfred Nobel (1896), Swedish inventor and founder of the prize.
- The 2024 laureate was Han Kang of South Korea, recognized for fiction confronting historical trauma and the fragility of life.
|
About Laszlo Krasznahorkai:
- Overview: Hungarian novelist celebrated for his dense, philosophical, and apocalyptic prose that examines the fragility of modern civilization.
- Background: Regarded as one of Europe’s leading postmodern writers, noted for long, flowing sentences and hypnotic rhythm.
- Themes & Style: His works probe moral collapse, spiritual decay, existential isolation, and the search for meaning amid disorder.
- Literary Voice: Combines dark humor with metaphysical reflection; often set in bleak, decaying landscapes where characters struggle between despair and artistic endurance.
- Recognition: Known as a “writer’s writer”, his art embodies a belief in the redemptive endurance of literature.
Major Works & Adaptations:
- Satantango (1985): Debut novel portraying a collapsing rural community; adapted by Béla Tarr into a seven-hour film, acclaimed for its realism and existential tone.
- The Melancholy of Resistance (1989): Allegory of hysteria and conformity in a small town; adapted as Werckmeister Harmonies (2000).
- War and War (1999): Follows a Hungarian archivist obsessed with preserving a manuscript symbolising human history; explores madness and transcendence.
- Seiobo There Below (2008): Interlinked stories on art and divinity across cultures; won the 2015 Man Booker International Prize.
- Baron Wenckheim’s Homecoming (2016): Tragicomic portrait of post-communist moral decay; won the 2019 National Book Award (Translated Literature).
Why in the News?
The 2025 Nobel Prize in Chemistry has been awarded to Richard Robson, Susumu Kitagawa, and Omar Yaghi for pioneering the creation of metal–organic frameworks (MOFs).

What are Metal–Organic Frameworks (MOFs)?
- Overview: They are crystalline materials composed of metal ions linked by organic molecules, forming a three-dimensional porous network capable of selectively trapping and storing gases, vapours, or liquids.
- Structure: Metal ions serve as nodes or connectors, while organic ligands (carbon-based linkers) create scaffold-like frameworks with very high surface area and controllable pore size.
- Porosity: MOFs possess some of the highest porosity among solids, often exceeding 7,000 square metres per gram, enabling the storage of large volumes of gases within minimal material.
- Flexibility: Organic linkers can be chemically modified, allowing custom design for specific interactions, such as selective gas capture or catalysis.
- Thermal and Chemical Stability: Advanced MOFs remain stable up to 300–400°C and can withstand diverse chemical environments, suitable for industrial and environmental use.
- Bonding Principle: Based on coordination chemistry, MOFs combine metal rigidity with organic flexibility, enabling precise control over molecular architecture.
- Functionality: Their open channels permit easy adsorption and desorption, making MOFs reusable, durable, and efficient for a range of scientific and industrial applications.
Applications of MOFs:
- Water Harvesting: Capture moisture from arid air and release it upon heating — enabling portable water generation in desert regions.
- Carbon Capture: Their selective pores allow efficient CO₂ capture and storage, aiding climate change mitigation.
- Hydrogen and Methane Storage: Act as solid sponges essential for fuel cells and clean energy systems.
- Pollutant Filtration: Remove PFAS (Per- and Polyfluoroalkyl Substances), heavy metals, and organic contaminants from water sources.
- Food Preservation: Absorb ethylene gas emitted by fruits, slowing ripening and extending shelf life.
- Catalysis and Sensing: Serve as heterogeneous catalysts and chemical sensors for trace-level detection in industrial settings.
- Clean Energy Systems: Integrated into batteries, fuel cells, and supercapacitors for energy storage due to high conductivity and surface area.
Scientific Development:
- Richard Robson (University of Melbourne, 1970s): He pioneered the idea of linking metal atoms and ligands into extended frameworks, though early models were fragile.
- Susumu Kitagawa (Kyoto University): Built porous coordination polymers, the first to demonstrate that gases could diffuse through molecular cavities—a defining MOF feature.
- Omar Yaghi (University of California, Berkeley, 1990s): Created robust, heat-resistant MOFs, standardised synthesis techniques, and coined the term “Metal–Organic Framework” in a 1995 Nature paper.
- Breakthrough Achievement: Yaghi’s team designed copper- and cobalt-based MOFs stable up to 350°C, capable of hosting guest molecules without collapse.
| [UPSC 2024] With reference to Direct Air Capture, an emerging technology, which of the following statements is/are correct?
I. It can be used as a way of carbon sequestration.
II. It can be a valuable approach for plastic production and in food processing.
III. In aviation, it can be a source of carbon for combining with hydrogen to create synthetic low-carbon fuel.
Select the correct answer using the code given below.
(a) I and II only (b) II only (c) I, II, and III* (d) None of the above statements is correct |
Why in the News?
The 2025 Nobel Prize in Physics has been awarded to John Clarke, Michel Devoret, and John Martinis for their discovery of macroscopic quantum mechanical tunnelling and energy quantisation in an electric circuit.

Discovery of Macroscopic Quantum Effects:
- Essence of the Discovery: John Clarke, Michel Devoret, and John Martinis proved that quantum effects—tunnelling and energy quantisation—can occur in macroscopic electrical circuits, not just in atoms or particles.
- Experiments (UC Berkeley, 1984–85): Demonstrated that superconducting circuits, visible to the naked eye, act as quantum systems when isolated from external disturbances.
- Observed Phenomena:
- Macroscopic Quantum Tunnelling: Electric current “jumps” through an insulating barrier even when classical physics predicts no flow.
- Energy Quantisation: The circuit holds only discrete energy levels, behaving like an artificial atom that exchanges energy in fixed quanta.
- Scientific Breakthrough: First experimental proof that quantum mechanics governs engineered large-scale systems, forming the foundation of quantum computing.
The Josephson Junction:
- Structure: Two superconductors separated by a thin insulating layer, allowing the passage of Cooper pairs paired electrons that move as a single quantum entity.
- Mechanism: Though insulators block current in classical systems, Cooper pairs tunnel through the barrier, producing a supercurrent without resistance.
- Key Berkeley Findings:
- The phase difference across the junction behaved as a quantum variable, showing discrete energy states.
- Spontaneous tunnelling of current produced measurable voltage, confirming macroscopic quantum tunnelling.
- Outcome: The Josephson junction became the first laboratory model of macroscopic quantum behaviour and the prototype for superconducting qubits used in today’s quantum computers.
Significance:
- Redefined Quantum Boundaries: Established that quantum laws are universal, applying from electrons to circuits of billions of atoms when quantum coherence is preserved.
- Foundation for Quantum Computing: Provided the conceptual basis for superconducting qubits, now central to Google, IBM, and TIFR quantum processors.
- Technological Impact: Enabled innovations in quantum sensors, precision metrology, and quantum communication through microwave-to-optical conversion.
- Philosophical Insight: Resolved the scale question of how large a system can remain quantum, proving that superconducting isolation preserves coherence even at macroscopic levels.
- Legacy: Bridged the quantum–classical divide, converting a theoretical boundary into experimentally verified reality, launching the modern quantum technology era.
| [UPSC 2022] Which one of the following is the context in which the term “qubit” is mentioned?
Options: (a) Cloud Services b) Quantum Computing* (c) Visible Light Communication Technologies (d) Wireless Communication Technologies |
Introduction
For decades, the immune system was viewed as a binary apparatus either attacking foreign invaders or remaining silent toward the body’s own cells. This year’s Nobel laureates, Mary Brunkow, Fred Ramsdell, and Shimon Sakaguchi, dismantled that simplistic view by uncovering the critical role of regulatory T-cells (Tregs) and the FOXP3 gene in maintaining self-tolerance. Their findings fundamentally redefined how scientists perceive immune regulation and opened the path for precision immunotherapy — one of modern medicine’s most promising frontiers.
The Science of Self-Tolerance: Why It’s in the News
The Nobel Committee’s recognition of research on regulatory T-cells (Tregs) and FOXP3 marks a watershed moment in immunology. For the first time, the prize acknowledges discoveries that explain how the immune system prevents itself from attacking the body. The work explains why autoimmune disorders like Type 1 diabetes, rheumatoid arthritis, and lupus occur when this “self-check” mechanism fails. It also connects molecular immunology to emerging therapies for cancer and transplantation. This is a landmark shift from viewing immunity as mere “defence” to seeing it as a balance of activation and restraint, a concept that has redefined global biomedical research.
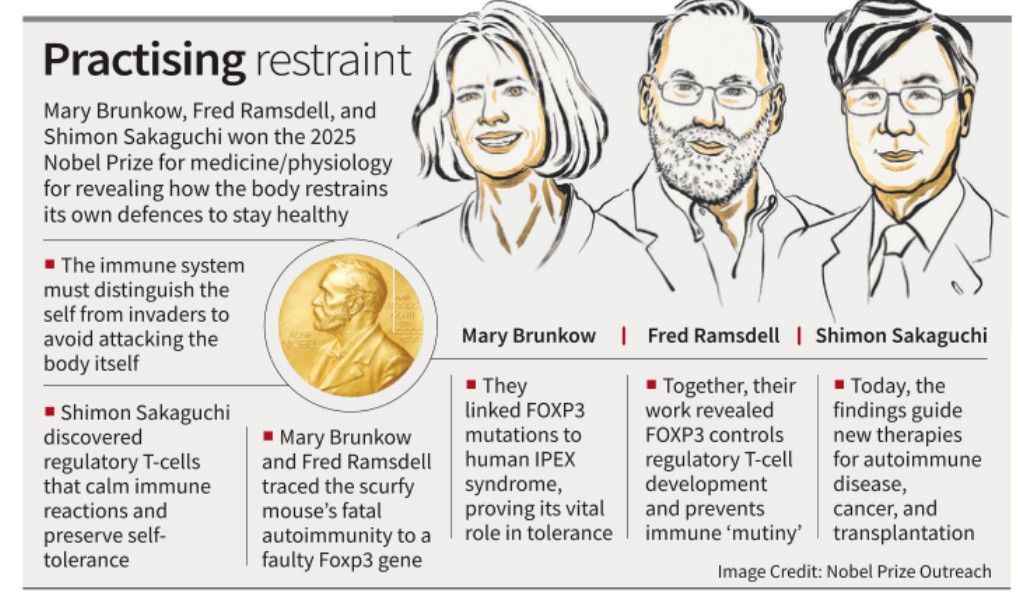
How the Nobel-winning Discovery Unfolded
- Early Understanding: In the 1990s, immunologists believed that self-reactive T-cells were deleted during their maturation. However, this could not explain why some autoreactive T-cells still existed in healthy people.
- Sakaguchi’s Breakthrough (1995): Identified a subset of CD4⁺ T-cells whose removal in mice led to multiple autoimmune disorders. Restoring them prevented disease — proving they act as regulators of immune overreaction.
- Discovery of FOXP3 Gene: Brunkow and Ramsdell, working in an industry lab (Celltech Chiroscience), traced severe autoimmune disease in male “scurfy” mice to a gene mutation on the X chromosome. They named it FOXP3.
- Human Correlation: Soon, mutations in FOXP3 were linked to lethal autoimmune syndromes in boys, confirming its pivotal role in human immune regulation.
How These Discoveries Transformed Immunology
- Redefining the Immune System: The immune system is now seen not as an on/off mechanism but as a dynamic ecosystem that balances activation (attack) with restraint (tolerance).
- New Therapeutic Frontiers:
-
- Autoimmune Diseases: Efforts are underway to expand or stabilise Tregs to curb harmful immune activation without broad immunosuppression.
- Transplant Medicine: Infusion of engineered Tregs improves graft acceptance and reduces rejection rates.
- Cancer Research: Selective depletion or reprogramming of tumour-associated Tregs enhances anti-tumour immunity without triggering autoimmunity.
From Lab to Life: The Translational Challenge
- Incremental Progress: Immunologists warn against overestimating breakthroughs. The immune system has multiple overlapping control layers, making clinical translation slow.
- High Cost Barrier: Cell-based therapies remain expensive, leading to inequitable access between high- and low-income populations.
- Ethical and Policy Dilemmas: Who gets access first? How do we regulate genetic manipulation or Treg engineering? These questions highlight the intersection of science, ethics, and public policy.
Private Sector and Scientific Innovation
- Industrial Discovery: The fact that Brunkow and Ramsdell made their discoveries in an industry setting (Celltech Chiroscience) underscores the potential of private-sector-led innovation in fundamental science.
- Public–Private Synergy: It reinforces how collaborations between academic research and biotech industry can accelerate discovery and application, a model India can emulate in its biotechnology policy framework.
Broader Implications for India and Global Health
- Indian Relevance: India’s growing burden of autoimmune diseases (such as lupus, celiac, and thyroiditis) highlights the need for indigenous immunogenetic research.
- Policy Perspective: Translating such research into affordable therapies aligns with National Biotechnology Development Strategy and Ayushman Bharat’s preventive healthcare goals.
- Global Impact: These discoveries open a new era of personalised immunotherapy, integrating molecular biology, bioethics, and equitable access.
Conclusion
The 2025 Nobel Prize reminds the world that progress in science often lies not in creating new weapons against disease but in understanding balance, the balance within nature and within ourselves. The discovery of Tregs and FOXP3 has rewritten textbooks, inspired therapies, and expanded our conception of what “self” and “immunity” truly mean. For policymakers and scientists alike, it represents the future, a fusion of molecular precision, ethical responsibility, and social justice.
PYQ Relevance
[UPSC 2021] The Nobel Prize in Physics of 2014 was jointly awarded to Akasaki, Amano and Nakamura for the invention of Blue LEDs in the 1990s. How has this invention impacted the everyday life of human beings?
Linkage: Both the 2014 Nobel for Blue LEDs and the 2025 Nobel for Treg–FOXP3 discovery represent paradigm shifts where scientific breakthroughs moved from lab theory to real-world transformation — the former revolutionised energy efficiency, while the latter is redefining human health and immune regulation.
Why in the News?
The Ramon Magsaysay Award 2025 has been awarded to Educate Girls, an Indian NGO working to promote girls’ education in rural and disadvantaged regions.
Other winners include:
- Shaahina Ali (Maldives): A noted environmental activist.
- Fr. Flaviano Antonio L. Villanueva (Philippines): A human rights defender, critic of Duterte’s drug war.
About Educate Girls:
- Founded as: Foundation to Educate Girls Globally; CEO: Gayatri Nair Lobo.
- Mission: Address gender inequality in education and uplift rural communities through girls’ schooling.
- Impact:
- Operates in India’s most rural and remote regions.
- Employs community workers (preraks, team balikas) to mobilise enrollment and retention.
- Creates ripple effects: education empowers girls → uplifts families → strengthens communities.
- Significance: It is the first Indian organisation to win the award since its inception in 1958.
About Ramon Magsaysay Award:
- “Nobel Prize of Asia”: Awarded annually since 1958.
- Purpose: Celebrate “greatness of spirit and transformative leadership” in Asia.
- Recognition: Individuals/organisations showing integrity in governance, service, and idealism in democracy.
- Origin:
- Established April 1957 by Rockefeller Brothers Fund trustees with support of the Philippines govt.
- Named in honour of Ramon Magsaysay, former Philippine President (1953–57), noted for administrative and military leadership.
- Original Categories (1958–2008): Govt Service, Public Service, Community Leadership, Journalism & Arts, Peace & International Understanding, and later Emergent Leadership (2001).
- Since 2009: Fixed categories dropped (except Emergent Leadership), award now honours diverse forms of excellence.
- Notable Indian Recipients:
- Vinoba Bhave (1958): Bhoodan movement.
- Mother Teresa (1962): humanitarian service.
- Satyajit Ray (1967): cinema.
- M.S. Subbulakshmi (1974): music.
- Arvind Kejriwal (2006): anti-corruption work.
- Ravish Kumar (2019): journalism.
- Sonam Wangchuk (2018): educational innovation.
- Educate Girls (2025): first Indian organisation to be honoured.
| [UPSC 2004] Sandeep Pandey, the winner of Ramon Magsaysay Award, is mainly an activist in:
Options: (a) a campaigner for urban sanitation (b) an anti-child labour activist (c) Environmental protection (d) Education and livelihood projects for Dalits* |
Why in the News?
Indian mathematician Dr. Rajula Srivastava has received the Maryam Mirzakhani New Frontiers Prize for her groundbreaking work in harmonic analysis and number theory.
About the Indian Laureate: Dr. Rajula Srivastava
- Affiliation: Hausdorff Center for Mathematics, University of Bonn and Max Planck Institute of Mathematics, Germany.
- Field of Work:
- Dr. Srivastava studied how to break down complex mathematical functions into simpler parts using advanced math tools.
- She worked on finding how close certain numbers can get to specific points on curved shapes in higher dimensions.
|
About the Maryam Mirzakhani New Frontiers Prize:
- Purpose: Recognises early-career women mathematicians (within two years of PhD completion) for outstanding research contributions.
- Named After: Maryam Mirzakhani — first woman and first Iranian to win the Fields Medal; known for her work on geometry and Riemann surfaces.
- Objective: To honour Mirzakhani’s legacy, encourage more women in mathematics, and elevate early-career contributions.
- Established: November 2019 by the Breakthrough Prize Foundation.
- Prize Amount: $50,000, may be shared by multiple recipients in a year.
- Eligibility: Women mathematicians who have:
- Recently completed their PhD (within 2 years)
- Shown exceptional promise and innovation in mathematical research.
Other Important Prizes in Mathematics:
- Fields Medal: Awarded every four years to mathematicians under 40, the Fields Medal is the most prestigious global mathematics award, established in 1936, and presented by the International Mathematical Union to recognize outstanding contributions to the field.
- Abel Prize: Instituted by the King of Norway in 2001, the Abel Prize is an annual international award recognizing lifetime achievement in mathematics, often considered the closest equivalent to the Nobel Prize in the discipline.
- Wolf Prize in Mathematics: Awarded annually since 1978 by the Wolf Foundation in Israel, the Wolf Prize honours exceptional achievements across all branches of mathematics and is ranked just below the Fields and Abel Prizes in prestige.
| [UPSC 2016] A recent movie titled The Man Who Knew Infinity is based on the biography of
Options: (a) S. Ramanujan* (b) S. Chandhrashekhar (c) S. N. Bose (d) C. V. Raman |
Why in the News?
Padma Awards 2025 recognised 139 individuals for their exceptional contributions across various fields. Notable awardees include actor S. Ajith Kumar, singer Pankaj Udhas, and cricketer Ravichandran Ashwin.

About Padma Awards:
- The Padma Awards are among India’s most prestigious civilian honors, announced annually on Republic Day’s eve.
- These awards recognize outstanding achievements in various fields, particularly public service.
- The GoI introduced the Bharat Ratna and Padma Vibhushan in 1954, with the three classes (Pahela Varg, Dusra Varg, and Tisra Varg) renamed in 1955 as Padma Vibhushan, Padma Bhushan, and Padma Shri.
- The awards are not titles and cannot be used as prefixes or suffixes to names.
- The number of awards given annually is capped at 120 (excluding posthumous awards and awards to NRIs/foreigners/OCIs).
Award Categories:
- Padma Vibhushan: For ‘exceptional and distinguished service.’
- Padma Bhushan: For ‘distinguished service of a high order.’
- Padma Shri: For ‘distinguished service.’
Eligibility and Nomination:
- Nominations are reviewed by the Padma Awards Committee, formed annually by the Prime Minister, with final approval from the President.
- Eligibility Criteria:
- Open to all individuals, regardless of race, occupation, position, or gender.
- Government employees, except doctors and scientists, are generally ineligible.
- Awards are generally not given posthumously, with exceptions in extraordinary cases.
- A recipient can be considered for a higher category only after five years since their last Padma award.
| [UPSC 2021] Consider the following statements in respect of Bharat Ratna and Padma Awards:
1. Bharat Ratna and Padma Awards are titles under Article 18(1) of the Constitution of India.
2. Padma Awards, which were instituted in the year 1954, were suspended only once.
3. The number of Bharat Ratna Awards is restricted to a maximum of five in a particular year.
Which of the above statements are not correct?
Options: (a) 1 and 2 only (b) 2 and 3 only (c) 1 and 3 only (d) 1, 2, and 3* |
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Nobel Prize in Economics

Why in the News?
The Nobel Memorial Prize in Economics for 2024 was awarded to Daron Acemoglu, Simon Johnson, and James A. Robinson for their research into why some nations are more prosperous than others.
What is the Nobel Economics Prize?
- Officially called the Bank of Sweden Prize in Economic Sciences in Memory of Alfred Nobel, established in 1968.
- It is not part of the original Nobel Prizes created by Alfred Nobel in 1895.
- Created by the Swedish central bank to honor Alfred Nobel’s legacy.
- Although not an original Nobel Prize, it is presented alongside the other Nobel Prizes on December 10, the anniversary of Nobel’s death.
- Includes a diploma, gold medal, and a one-million-dollar prize for the laureates.
|
This Year’s Nobel for Economics:
- The 2024 Nobel awardees’ research focused on the role of societal institutions in determining a country’s economic prosperity.
- The laureates distinguished between inclusive institutions, which promote democracy, rule of law, and protection of property rights, and extractive institutions, where power is concentrated and resources are exploited for the benefit of a few.
- Their study explains how inclusive institutions foster long-term economic growth by providing stability and incentives, while extractive institutions undermine growth by discouraging investment and innovation.
- The researchers also explored how European colonization shaped current economic disparities by establishing either inclusive or extractive institutions, with decisions influenced by factors such as mortality risks during colonization.
Significance of the study
- The study highlights the critical role of societal institutions in determining a country’s prosperity.
- It distinguishes between inclusive institutions that foster growth and extractive institutions that hinder it.
- It explains how colonial history shaped modern economic disparities.
- The research offers policy insights for building inclusive institutions to promote long-term prosperity.
- It has global implications for reducing inequality and promoting sustainable development.
PYQ:
[2019] The long-sustained image of India as a leader of the oppressed and marginalised nations has disappeared on account of its new found role in the emerging global order.’ Elaborate. |
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Nobel Prize of Literature

Why in the News?
The 2024 Nobel Prize has been awarded to South Korean writer Han Kang for her “intense poetic prose that confronts historical traumas and exposes the fragility of human life.”
Who is Han Kang?
- Han Kang is a South Korean writer born in 1970 in Gwangju, South Korea.
- She is known for her poetic and experimental writing style that confronts themes of historical trauma, violence, grief, and the fragility of human life.
- After starting her literary career with poetry, she became widely recognized for her novels that address complex human emotions and the consequences of social and political structures.
Her Literary Works:
- The Vegetarian (2007): This novel, which won the Man Booker International Prize in 2016, revolves around a woman who decides to stop eating meat and faces extreme reactions from her family. The story explores themes of control, independence, and violence. It was her breakthrough novel, translated into English in 2015.
- Human Acts (2016): Set against the backdrop of the 1980 Gwangju Uprising, this novel tells the story of the massacre of protesting students by the South Korean military. Han Kang gives voice to the victims of history with a style that is both experimental and visionary.
- The White Book (2017): An elegy dedicated to a sibling who passed away shortly after birth. The novel is a meditation on grief, narrated through white objects, symbolizing loss and memory.
- Greek Lessons (2023): This novel, first published in Korean in 2011, tells the story of a woman who loses her ability to speak and her teacher, who is losing his sight. The novel explores loss, intimacy, and the connection between language and identity.
- We Do Not Part (2025, forthcoming): This upcoming novel focuses on two women confronting a hidden massacre in Korean history from the 1940s. It explores how trauma can be transformed into art.
Citation for Nobel Prize 2024
- The Swedish Academy’s official bio-bibliography highlights her ability to create universal narratives using a poetic and radical imagination.
- Han Kang is praised for addressing patriarchy, violence, and historical injustices, while also examining the connections between body and soul and the living and the dead.
- The academy notes that she has become an innovator in contemporary prose, showing how literature can speak the truth through her powerful and experimental style.
Recent Nobel Prizes in Literature:
- 2023: Jon Fosse (Norway) for his innovative minimalistic plays and prose, focusing on the human condition.
- 2022: Annie Ernaux (France) for her courageous exploration of personal and collective memory.
- 2021: Abdulrazak Gurnah (Tanzania) for his compassionate depictions of colonialism and migration.
- 2020: Louise Gluck (USA) for her deeply personal poetry that resonates universally.
| Rabindranath Tagore: Won the Nobel Prize in 1913 for “Gitanjali,” becoming the first non-European laureate in Literature, recognized for his sensitive and spiritual poetry. |
|
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Artificial Neural Network, Physics Nobel, 2024
Why in the News?
John Hopfield and Geoffrey Hinton have been awarded the Nobel Prize in Physics, 2024 for their pioneering work that laid the foundation for machine learning and artificial neural network.

What is Artificial Neural Network?
- Artificial neural networks are systems inspired by the structure of the brain.
- In these networks, nodes represent the brain’s neurons, and they influence each other through connections, similar to synapses in the brain.
- These connections can be made stronger or weaker through training.
- For instance, connections between nodes with high values can become stronger as the network learns.
- Since the 1980s, Hopfield and Hinton have been conducting important research on artificial neural networks.
|
Significance of their contribution
- Hinton’s work on the Boltzmann machine and other neural networks helped drive the explosive growth of machine learning.
- His methods continue to play a significant role in modern AI, enabling technologies like image classification and pattern recognition.
PYQ:
[2020] With the present state of development, Artificial Intelligence can effectively do which of the following?
1 Bring down electricity consumption in industrial units
2. Create meaningful short stories and songs
3. Disease diagnosis
4. Text-to-Speech Conversion
5. Wireless transmission of electrical energy
Select the correct answer using the code given below:
(a) 1, 2, 3 and 5 only
(b) 1, 3 and 4 only
(c) 2, 4 and 5 only
(d) 1, 2, 3, 4 and 5 |
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Initiative related to scientific research;
Why in the News?
In 2023, the government replaced numerous opaque science awards with the Rashtriya Vigyan Puraskar (RVP), a streamlined set with clear criteria. It aimed to address concerns about transparency by involving committees led by respected scientists.
Introduction of Rashtriya Vigyan Puraskar (RVP):
- In 2023, the government replaced multiple science-related awards with the RVP, a consolidated and streamlined set of awards.
- RVP introduced specific categories and clear eligibility criteria to enhance transparency and reduce opacity in the selection process.
- Committees led by reputed scientists were tasked with finalizing the awardees, a measure intended to address concerns about bureaucratic interference.
|
Concerns Over Selection Process:
- Chance of political interference: The modification of rules overnight and the omission of some scientists from the final list, despite being shortlisted by the RVP committee, created suspicion of political interference.
- Omission of the critic of Govt policy: Notably, some of the omitted scientists had previously criticized government policies, leading to concerns that the awards process might be used to marginalize dissenting voices within the scientific community.
Shift in Authority:
- Original Process: Initially, the RVP committee head had the authority to finalize the awardees.
- Recent Change: A last-minute rule modification allowed the committee head to only “recommend” the final list to the Science Ministry, increasing the possibility of government intervention in the selection process.
- Recognition Framework: The RVP awards are categorized into four main types: Vigyan Ratna for lifetime achievements, Vigyan Shri for distinguished contributions, Vigyan Yuva for young scientists, and Vigyan Team for collaborative efforts.
Impact on the Scientific Community
- Concerns Over Transparency: The modification of selection criteria just before the announcement of awardees has led to suspicions that political motives might influence who receives recognition.
- Notably, some scientists who were initially included in the recommended list were omitted from the final public announcement, prompting questions about whether political pressures were involved.
- Exclusion of Dissenting Voices: Many excluded scientists had previously expressed dissent against government policies. This trend suggests a troubling pattern where recognition is contingent upon alignment with governmental perspectives, potentially stifling academic freedom and critical discourse within the scientific community.
Way forward:
- Strengthen Transparency and Autonomy: Establish an independent, non-governmental oversight body to ensure that the selection process remains transparent and free from political interference.
- Safeguard Academic Freedom: Implement safeguards to prevent exclusion based on political or ideological views, ensuring that recognition is purely merit-based.
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Winners at Olympics, 2024
Why in the News?
India has won a total of 6 medals at the 33rd Summer Olympic Games in Paris, with 5 bronze medals and 1 silver medal.
Full List of Medal Winners
|
Game |
Medal |
Details |
| Manu Bhaker |
10m Air Pistol Women’s |
Bronze |
Secured a bronze medal with a score of 221.7 after 22 attempts.
|
| Manu Bhaker, Sarabjot Singh |
10m Air Pistol Mixed Team |
Bronze |
It was a team event.
Manu now is the first Indian woman to win two medals in a single Summer Games. |
| Swapnil Kusale |
50m Rifle 3 Position |
Bronze |
First-ever Indian medal in this event. |
| Men’s Hockey Team |
Hockey |
Bronze |
Defeated Spain by 2-1 in the third-place playoff, with skipper Harmanpreet Singh scoring both goals. |
| Neeraj Chopra |
Men’s Javelin Throw |
Silver |
Secured medal with a distance of 89.45m.
Became first Indian to win two Olympic medals in track-and-field. |
| Aman Sehrawat |
Men’s Wrestling Freestyle 57kg |
Bronze |
Men’s Freestyle 57kg category by defeating Darian Toi Cruz of Puerto Rico 13-5. |
PYQ:
[2021] Consider the following statements in respect of the Laureus World Sports Award which was instituted in the year 2000 :
1. American golfer Tiger Woods was the first winner of this award.
2. The award was received mostly by ‘Formula One’ players so far.
3. Roger Federer received this award maximum number of times compared to others.
Which of the above statements are correct?
(a) 1 and 2 only
(b) 2 and 3 only
(c) 1 and 3 only
(d) 1, 2 and 3 other |
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Kavli Prize
Why in the News?
The winners of the 2024 Kavli Prize were announced, recognising achievements in astrophysics, neuroscience, and nanoscience.
About Kavli Prize
- The Kavli Prize celebrates outstanding achievements in astrophysics, neuroscience, and nanoscience.
- It is established in honour of Norwegian-American businessman and philanthropist Fred Kavli (1927-2013).
- Awarded biennially, the prize recognizes researchers whose work pushes the boundaries of human knowledge and enhances our understanding of the universe, the brain, and nanoscale phenomena.
About Fred Kavli
- Fred Kavli, born in Erejsford, Norway, immigrated to California in 1956 and quickly made a mark in the engineering field.
- His entrepreneurial journey began in 1958 with the founding of Kavlico, a pioneering company known for its precision pressure sensors used across diverse industries, from aviation to consumer electronics.
The Kavli Foundation:
- In 2000, after selling Kavlico for $340 million, Fred Kavli established the Kavli Foundation.
- This philanthropic organisation supports fundamental research aimed at improving global quality of life.
- The foundation operates 20 institutes worldwide dedicated to astrophysics, neuroscience, nanoscience, and theoretical physics.
|
Prize Structure and Prestige:
- Comparison with Nobel Prize: The Kavli Prize mirrors the Nobel Prize in its prestige and international recognition but differs by not limiting awards to achievements within the preceding year, allowing broader scope and longevity in selection criteria.
- Award Ceremony: Each Kavli Prize includes a $1 million cash award per field, a scroll, and a 7 cm diameter medal. The ceremony, held at the Oslo Concert Hall and attended by global scientific leaders, features a red-carpet event, emphasizing its significance in the scientific community.
- Selection Process:
- Committees: Three international committees nominate and review candidates, providing a unanimous recommendation to the Norwegian Academy of Science and Letters.
- Nominees: Nominees come from prestigious institutions worldwide, ensuring a diverse and comprehensive evaluation process.
Winners of the 2024 Kavli Prize:
| Field |
Winners |
Contributions |
| Astrophysics |
David Charbonneau (Harvard University), Sara Seager (Massachusetts Institute of Technology) |
Pioneering methods for detecting atomic species in planetary atmospheres and measuring their thermal infrared emission, are crucial for identifying molecular fingerprints in atmospheres of exoplanets. |
| Nanoscience |
Robert Langer (MIT), Armand Paul Alivisatos (University of Chicago), Chad Mirkin (Northwestern University) |
Langer: Nano-engineering for controlled drug delivery systems.
Alivisatos: Development of semiconductor quantum dots for bio-imaging.
Mirkin: Concept of spherical nucleic acids (SNAs) for applications in gene regulation and immunotherapy. |
| Neuroscience |
Nancy Kanwisher (MIT), Winrich Freiwald (Rockefeller University), Doris Tsao (University of California, Berkeley) |
Mapping brain functions related to facial recognition using neuroimaging and neuronal recording techniques, identifying brain centers and neural architectures involved in face processing. |
PYQ:
[2021] The Nobel Prize in Physics of 2014 was jointly awarded to Akasaki, Amano and Nakamura for the invention of Blue LEDs in the 1990s. How has this invention impacted the everyday life of human beings?
[2018] Discuss the work of ‘Bose-Einstein Statistics’ done by Prof. Satyendra Nath Bose and show how it revolutionized the field of Physics.
[2008] Nobel Prize-winning scientist James D. Watson is known for his work in which area? (2008)
(a) Metallurgy
(b) Meteorology
(c) Environmental protection
(d) Genetics |
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Longitude Prize, AMR
Why in the News?
Sysmex Astrego, a Sweden-based diagnostic company, won the £10 million Longitude Prize for developing a high-tech UTI (Urinary Tract Infection) test called the PA-100 AST System.
What is Antimicrobial Resistance?
- Antimicrobial Resistance (AMR) occurs when bacteria, viruses, fungi, and parasites change so that medicines no longer work against them.
- This makes infections harder to treat and can lead to more severe illness and even death.
- AMR can occur naturally through genetic changes or when one type of microorganism picks up resistance from another.
- It can also occur randomly due to mutations or when resistant genes spread between different organisms.
- The main reasons for AMR are antibiotics incorrectly or too often, not having access to clean water and good hygiene, not doing enough to prevent infections, and people not knowing how to use antibiotics properly.
Global Impact of AMR
- Global Threat: Antibiotic-resistant infections caused nearly 1.3 million deaths in 2019, with projections to exceed cancer-related deaths by 2050.
- Economic Impact: The global economy faces potential losses of up to 4 trillion by 2030 and 100 trillion by 2050 due to the AMR crisis.
|
About the Longitude Prize
- The Longitude Prize, valued at £8 million (₹85 crore), was recently awarded for its profound implications in combating antimicrobial resistance (AMR).
- The prize launched in 2014, aims to revolutionize infection management globally.
- Historical Context:
- Origins: Established in 1714 to solve the maritime longitude problem, demonstrating the critical need for accurate navigation.
- Modern Application: The 21st-century Longitude Prize addresses pressing global challenges like AMR, incentivizing innovative solutions.
- Goal of the Prize:
- Aims to replace current lab-based diagnostic methods with rapid tests that guide targeted antibiotic therapies, combating AMR effectively.
About PA-100 AST System
- Technology Breakthrough: Developed by Sysmex Astrego, this system uses a phenotypic test that identifies UTI bacteria and performs antibiotic susceptibility testing in under 45 minutes.
- Operation: A single-use cartridge processes less than half a milliliter of urine, using microfluidic traps and phase-contrast imaging in a shoe box-sized reader instrument.
- Clinical Impact: Provides rapid “sensitive” or “resistant” results for antibiotics, aiding doctors in immediate, accurate treatment decisions at the point of care.
Current Challenges in UTI Diagnosis
- Limited Accuracy: Current options include urine dipstick tests for quick results but limited accuracy (50-60%) and lab cultures with 2-3 day turnaround times.
- Impact on Treatment: Delayed diagnosis often leads to empirical antibiotic use, contributing to antibiotic resistance worldwide.
Significance for India
- Healthcare Burden: In India, millions seek treatment for UTIs annually, impacting lives and healthcare systems significantly.
- Potential: Rapid, accurate point-of-care tests could revolutionize UTI management globally, saving lives and reducing healthcare costs.
PYQ:
[2014] Can overuse and free availability of antibiotics without a Doctor’s prescription be contributors to the emergence of drug-resistant diseases in India? What are the available mechanisms for monitoring and control? Critically discuss the various issues involved.
[2019] Which of the following are the reasons for the occurrence of multi-drug resistance in microbial pathogens in India?
- Genetic predisposition of some people
- Taking incorrect doses of antibiotics to cure diseases
- Using antibiotics in livestock farming
- Multiple chronic diseases in some people
Select the correct answer using the code given below.
(a) 1 and 2
(b) 2 and 3 only
(c) 1, 3 and 4
(d) 2, 3 and 4 |
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Padma Awards
Mains level: NA

Why in the news?
The President of India, Smt. Droupadi Murmu presented 3 Padma Vibhushan, 8 Padma Bhushan and 55 Padma Shri Awards for the year 2024.
About Padma Awards
- The Padma Awards are among India’s most prestigious civilian honors.
- It is announced annually on Republic Day, following the Bharat Ratna.
- These awards are designed to recognize outstanding achievements across various fields and disciplines, particularly those involving public service.
- It was introduced in 1954 along with Bharat Ratna.
- Initially, the Padma Vibhushan had three classes: Pahela Varg, Dusra Varg, and Tisra Varg, which were renamed in 1955 as Padma Vibhushan, Padma Bhushan, and Padma Shri, respectively.
Award Categories
There are three categories of Padma Awards:
- Padma Vibhushan: This is awarded for ‘exceptional and distinguished service.’
- Padma Bhushan: This is for ‘distinguished service of a high order.’
- Padma Shri: This is given for ‘distinguished service.’
Eligibility Criteria
- The awards are open to all individuals regardless of race, occupation, position, or gender.
- The awards are usually not given posthumously, but exceptions can be made in extraordinary cases.
- A recipient can be considered for a higher category of Padma award only if at least five years have passed since their previous Padma honor. However, exceptions can be made for exceptionally deserving cases.
- NOT ELIGIBLE: Government employees, except doctors and scientists.
- The awards are not titles and cannot be used as prefixes or suffixes to the awardees’ names.
- The number of awards given annually (excluding posthumous awards and those to NRIs/foreigners/OCIs) is capped at 120.
Nomination Process
- Nominations are reviewed by the Padma Awards Committee, formed annually by the Prime Minister.
- The Committee, led by the Cabinet Secretary, includes the Home Secretary, Secretary to the President, and 4-6 distinguished members.
- The Committee’s recommendations are forwarded to the Prime Minister and the President for final approval.
Dignitaries of the Padma Awards 2024
|
Recipient (Occupation/Field) |
| Padma Vibhushan |
- M. Venkaiah Naidu (Former Vice-President)
- Bindeshwar Pathak (posthumously) (Founder of Sulabh International)
- Padma Subrahmanyam (Bharatanatyam dancer)
|
| Padma Bhushan |
- Mithun Chakraborty (Actor)
- Usha Uthup (Singer)
- Ram Naik (Former Governor of Uttar Pradesh)
- Sitaram Jindal (Industrialist)
- Tejas Madhusudan Patel (Cardiologist)
- Dattatray Ambadas Mayaloo (Rajdutt) (Marathi film director)
- Chandreshwar Prasad Thakur (Physician)
|
| Padma Shri |
- Rohan Bopanna (Tennis player)
- Khalil Ahamad (Master carpet weaver)
- Kaluram Bamaniya (Folk singer)
- Rezwana Choudhury Bannya (Bangladeshi singer)
- Naseem Bano (Chikankari embroidery artist)
- Gita Roy Barman (Rajbongshi folk singer)
- Chitta Ranjan Debbarma (Spiritual figure from Tripura)
- Sarbeswar Basumatary (Tribal farmer from Assam)
|
PYQ:
[2021] Consider the following statements in respect of the Bharat Ratna and Padma Awards.
- Bharat Ratna and Padma Awards are titled under Article 18(1) of the Constitution of India.
- Padma Awards, which were instituted in the year 1954, were suspended only once.
- The number of Bharat Ratna Awards is restricted to a maximum of five in a particular year.
Which of the above statements is not correct?
(a) 1 and 2 only
(b) 2 and 3 only
(c) 1 and 3 only
(d) 1, 2 and 3 |
https://pib.gov.in/PressReleaseIframePage.aspx?PRID=2018533
Note4Students
From UPSC perspective, the following things are important:
Prelims level: CIDC Vishwakarma Awards , CSR
Mains level: NA
Why in the news?
SJVN Limited has won two prestigious awards at the 15th Construction Industry Development Council (CIDC) Vishwakarma Awards 2024, for their Corporate Social Responsibility (CSR) initiatives.
About CIDC Vishwakarma Awards
- The CIDC Vishwakarma Awards is one of the most esteemed recognitions within the construction sector.
- It was launched in the year 2005.
- The awards are named after Vishwakarma, the divine architect and engineer in Hindu mythology, symbolizing craftsmanship, creativity, and skill in construction.
Key details about the Award
- Organizer: The awards are organized by the Construction Industry Development Council (CIDC), which is a body established by the GoI to promote the construction industry’s development and growth.
- Categories: The awards cover a wide spectrum of categories, including:
-
- Construction Projects: Recognizing outstanding projects across different sectors such as residential, commercial, infrastructure, and industrial construction.
- Construction Technologies: Honoring innovative technologies and techniques that enhance construction processes, efficiency, and sustainability.
- Construction Equipment: Acknowledging advancements in construction machinery, tools, and equipment.
- Health, Safety, and Environment: Recognizing initiatives and practices that prioritize worker safety, environmental protection, and sustainability in construction.
- Individual Achievements: Celebrating the contributions of professionals and leaders who have made significant impacts in the construction industry.
- Others: Additional categories may include awards for sustainability, CSR initiatives, and emerging trends in construction.
PYQ:
[2020] In rural road construction the use of which of the following is preferred for ensuring environmental sustainability or to reduce carbon footprint?
1. Copper slag
2. Cold mix asphalt technology
3. Geotextiles
4. Hot mix asphalt technology
5. Portland cement
Select the correct answer using the code given below:
(a) 1, 2 and 3 only
(b) 2, 3 and 4 only
(c) 4 and 5 only
(d) 1 and 5 only |
Back2Basics: Corporate Social Responsibility (CSR)
|
Description |
| What is it? |
Self-regulating business model for social and environmental impact. |
| Regulation in India |
Mandated under Companies Act, 2013 (amendment in 2014). |
| Investment Areas |
Promote rural development, healthcare, education, environment, etc. |
| CSR Committee |
Mandatory for companies meeting Rs 500 Cr net worth or Rs 1000 Cr turnover criteria. |
| Spending Requirement |
At least 2% of average net profits of the last three financial years. |
| Applicability Criteria |
Net worth >= Rs 500 Cr, Turnover >= Rs 1000 Cr, or Net profit >= Rs 5 Cr. |
| Adjustment for New Cos. |
Use average net profits of preceding years to calculate spending. |
| Applicability Period |
Applies before the completion of three financial years for companies. |
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Padma Awards
Mains level: Read the attached story

Central Idea
- Former Vice-President M. Venkaiah Naidu, actors Chiranjeevi and Vyjayanthimala Bali, and Bharatanatyam dancer Padma Subrahmanyam will be conferred with Padma Vibhushan, the country’s second-highest civilian award, the Central government announced on the eve of Republic Day.
- Bindeshwar Pathak, founder of Sulabh International, will be conferred with the award posthumously.
About Padma Award
- The Padma Awards are among India’s most prestigious civilian honors, announced annually on Republic Day’s eve, following the Bharat Ratna.
- These awards are designed to recognize outstanding achievements across various fields and disciplines, particularly those involving public service.
- The Government of India introduced the Bharat Ratna and Padma Vibhushan in 1954.
- Initially, the Padma Vibhushan had three classes: Pahela Varg, Dusra Varg, and Tisra Varg, which were renamed in 1955 as Padma Vibhushan, Padma Bhushan, and Padma Shri, respectively.
Award Categories
There are three categories of Padma Awards:
- Padma Vibhushan: This is awarded for ‘exceptional and distinguished service.’
- Padma Bhushan: This is for ‘distinguished service of a high order.’
- Padma Shri: This is given for ‘distinguished service.’
Eligibility Criteria
- The awards are open to all individuals regardless of race, occupation, position, or gender.
- Government employees, except doctors and scientists, are generally ineligible.
- The awards are usually not given posthumously, but exceptions can be made in extraordinary cases.
- A recipient can be considered for a higher category of Padma award only if at least five years have passed since their previous Padma honor. However, exceptions can be made for exceptionally deserving cases.
Nomination Process
- Nominations are reviewed by the Padma Awards Committee, formed annually by the Prime Minister.
- The Committee, led by the Cabinet Secretary, includes the Home Secretary, Secretary to the President, and 4-6 distinguished members.
- The Committee’s recommendations are forwarded to the Prime Minister and the President for final approval.
Limitations
- The awards are not titles and cannot be used as prefixes or suffixes to the awardees’ names.
- The number of awards given annually (excluding posthumous awards and those to NRIs/foreigners/OCIs) is capped at 120.
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Karpoori Thakur
Mains level: NA

Introduction
- On January 23, the Indian government made a significant announcement, posthumously awarding the Bharat Ratna, India’s highest civilian honor, to Karpoori Thakur, the former chief minister of Bihar.
- Thakur, affectionately known as ‘Jannayak’ or the leader of the people, leaves behind a legacy of enduring policy decisions that continue to shape Bihar and the nation.
Karpoori Thakur: Life and Career
- A Leader of Contrasts: Thakur, born on January 24, 1924, in Bihar, emerged as the foremost backward caste leader in the state, despite hailing from the minority nai (barber) caste.
- Political Activism: He actively participated in the freedom struggle, enduring imprisonment for his convictions.
- Decades of Public Service: Thakur served as an MLA from 1952 until his passing in 1988, except for a brief period when he became an MP in 1977.
- Respected Clean Image: Despite holding public office for three decades, Thakur maintained a clean image and refrained from personal enrichment through government resources.
Landmark Policy Decisions
- Education Reform: Removed English as a compulsory subject for matriculation examinations, aiming to make education more accessible.
- Prohibition of Alcohol: Enforced a ban on alcohol consumption to address societal issues stemming from alcohol abuse.
- Job Opportunities for Engineers: Introduced preferential treatment for unemployed engineers in government contracts, leading to employment for approximately 8,000 engineers.
- Layered Reservation System: Implemented the ‘Karpoori Thakur Formula’ in June 1970, providing 26% reservation, including 12% for OBCs, 8% for economically backward OBCs, 3% for women, and 3% for economically disadvantaged individuals from upper castes.
Challenges and Consequences
- Immediate Opposition: Thakur’s reservation policy faced significant opposition, especially from upper castes, and his government fell due to its implementation.
- Polarization and Derogatory Remarks: His caste background led to derogatory slogans and polarization in society, despite his clean governance record.
- Political Fallout: Thakur’s decision to prioritize the reservation policy was perceived by some as hasty, unlike leaders who consolidated their positions before introducing similar policies.
Karpoori Thakur’s Enduring Legacy
- Mandal-Kamandal Politics: Thakur’s legacy remains influential in the ongoing Mandal-Kamandal political landscape.
- Recognition by Leaders: Bihar CM Nitish Kumar, himself from a numerically small EBC community, has closely associated himself with Thakur’s legacy.
- Political Continuation: Thakur’s policies continue to shape the agendas of political parties in Bihar, highlighting the lasting impact of his commitment to social justice.

Central idea
Claudia Goldin’s Nobel Prize win highlights the belated acknowledgment of gender dynamics in labor markets, prompting a reevaluation of entrenched biases in economics. Feminist economists stress the imperative to dismantle androcentric biases, advocating for a more inclusive economic theory that reflects diverse experiences.
Key Terms for quality answers:
- Androcentric biases
- Economic man
- Gender inequalities
- Unpaid work
- Masculinity in economics
- Empirical findings
- Feminist economists
- Social mechanisms
Key Phrases for improving mains score:
- Androcentric Biases: Are gender-based prejudices or preferences that favor male perspectives, often manifested in economic theories that reflect traditional gender roles and reinforce a male-centric viewpoint.
- Economic Man: Is a theoretical construct representing a rational, self-interested individual in economic models. It simplifies human behavior for analytical purposes but is critiqued for its failure to capture the complexities of real-life decision-making.
- Humanizing Economics: Involves infusing empathy, emotions, and a more realistic understanding of human behavior into economic analyses, recognizing that individuals are not solely motivated by rational self-interest.
Key Highlights:
- Claudia Goldin wins Nobel Prize in Economic Sciences for gender dynamics research in labor markets.
- Recognition prompts reflection on the delayed acknowledgment of gender-focused economic research.
- Economics traditionally male-dominated, leading to the marginalization of gender inequality issues.
Challenges:
- Under-representation of women in economics.
- Androcentric biases in economic theories, perpetuating gender hierarchies.
- Economic models ignoring gendered experiences and unpaid work, especially by women.
- Limited understanding of non-market spaces like households, hindering accurate economic analysis.
- Economic man assumptions perpetuate gender stereotypes and fail to question existing hierarchies.
- Masculinity in economics detaches the discipline from gendered experiences, particularly of women.
Analysis:
- Feminist economists call for an economic theory free of androcentric biases to address gender inequalities.
- Economic models fail to account for the contributions of women as unpaid workers, impacting the accuracy of empirical findings.
- Biases in economic theory can affect statistical methods and interpretation of empirical results.
- Economic rationality may overlook social mechanisms, leading to misinterpretation of empirical findings.
Way Forward:
- Educational Initiatives: Propose educational programs to sensitize economists to gender biases and promote inclusivity.
- Policy Changes: Advocate for policy changes within academic institutions to encourage diverse perspectives in economic research.
- Recognizing Diverse Contributions: Encourage acknowledgment of the work of economists from diverse backgrounds.
- Inclusive Policies: Advocate for policies that actively promote diversity and inclusivity within economics departments.
- Training Economists: Suggest incorporating training on mixed methods in economics education.
- Interdisciplinary Collaboration: Promote collaboration with sociologists, anthropologists, and other disciplines to enrich economic research
Conclusion:
Claudia Goldin’s Nobel Prize win serves as a catalyst for a much-needed evolution in economic thinking. By addressing historical biases, overcoming gender-based challenges, and embracing a more inclusive and nuanced approach, the discipline can truly reflect the complexities of reality.
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Ramon Magsaysay Award
Mains level: Not Much

Central Idea
- Dr. R. Ravi Kannan, a surgical oncologist and Director of Cachar Cancer Hospital and Research Centre (CCHRC) in Assam, has been named one of the 2023 Ramon Magsaysay Awardees.
- His pioneering efforts have revolutionized cancer treatment in Assam by prioritizing people-centric and pro-poor healthcare solutions.
Dr. Kannan’s Inspiring Work
- Under Dr. Kannan’s guidance, CCHRC evolved from having limited facilities to encompassing 28 departments, including oncology, pathology, radiology, microbiology, epidemiology, and palliative care.
- The hospital introduced pro-poor measures such as free treatment, lodging, meals, ad hoc employment for caregivers, and a homecare program to address patients’ challenges in continuing treatment due to poverty and distance.
- The hospital team extended their support by providing training to family members in pain management and palliative care, and distributing free medicines.
- As a result, the compliance rate for cancer treatment increased remarkably, from 28% to 70%.
About Ramon Magsaysay Award
- The Ramon Magsaysay Award, established in 1958, stands as a significant accolade, often referred to as Asia’s equivalent of the Nobel Prize.
- This prestigious award honors individuals and organizations in Asia for their exceptional contributions to society across various domains.
Who was Ramon Magsaysay?
- Ramon Magsaysay was the Philippines’ president from 1953 until his tragic death in a plane crash in 1957.
- He gained prominence during World War II when Japanese forces occupied the Philippines, then a US colony.
- In December 1953, he was elected president from the Nationalist Party, the country’s oldest political party.
- Post-war chaos gripped the Philippines in 1946, accompanied by a widening gap between the rich and poor, exacerbated by the expansion of capitalism.
- Amidst suspicions of communist affiliations and demands for peasant rights, leaders were targeted by the government, aligned with the USA.
- Magsaysay’s administrative and military strategies played a pivotal role in countering the perceived threat of communism.
Indian Awardees: A Legacy of Excellence
Eminent Indians have been recognized through the Ramon Magsaysay Award:
- Vinoba Bhave (1958)
- Mother Teresa (1962)
- Kamaladevi Chattopadhyay (1966)
- Satyajit Ray (1967)
- Mahasweta Devi (1997)
- Arvind Kejriwal (2006)
- Anshu Gupta of Goonj (2015)
- Bezwada Wilson, human rights activist (2016)
- Ravish Kumar, journalist (2019)
Significance of the Award
- The Ramon Magsaysay Award symbolizes a deep commitment to altruism and service, acknowledging outstanding contributions that make a positive impact on society.
- It is a reminder that individuals and organizations can effect transformative change through their selfless efforts.
Note4Students
From UPSC perspective, the following things are important:
Prelims level: CR Rao
Mains level: Not Much

Central idea: Indian-American statistician Calyampudi Radhakrishna Rao has been awarded the 2023 International Prize in Statistics, which is considered the Nobel Prize for statistics. He is 102 YO.
Who is C. R. Rao?
- R. Rao, is an Indian-American mathematician and statistician.
- He is currently professor emeritus at Pennsylvania State University and Research Professor at the University at Buffalo.
- Rao has been honoured by numerous colloquia, honorary degrees, and festschrifts and was awarded the US National Medal of Science in 2002.
- The American Statistical Association has described him as “a living legend whose work has influenced not just statistics, but has had far reaching implications for fields as varied as economics, genetics, anthropology, geology, national planning, demography, biometry, and medicine.”
- The Times of India listed Rao as one of the top 10 Indian scientists of all time.
Rao’s Groundbreaking Paper
- The research paper, “Information and accuracy attainable in the estimation of statistical parameters,” was published in 1945 in the Bulletin of the Calcutta Mathematical Society.
- The paper provided a lower limit on the variance of an unbiased estimate for a finite sample, which has since become a cornerstone of mathematical statistics.
Key outcomes of his research
Rao’s 1945 paper has three outcomes-
- Cramer-Rao inequality: It provides a lower limit on the variance of an unbiased estimate for a finite sample.
- Rao-Blackwell Theorem: It provides a method to improve an estimate to an optimal estimate.
- Information geometry: It is a new interdisciplinary area called “information geometry,” which integrated principles from differential geometry into statistics, including the concepts of metric, distance, and measure.
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Abel Prize
Mains level: Not Much

The Abel Prize for mathematics for 2023 was awarded to Argentine-American Luis Caffarelli, an expert in “partial differential equations” which can explain phenomena ranging from how water flows to population growth.
Abel Prize
- The Abel Prize is a prize awarded annually by the King of Norway to one or more outstanding mathematicians.
- It is named after Norwegian mathematician Niels Henrik Abel (1802–1829) and directly modeled after the Nobel Prizes.
- It comes with a monetary award of 7.5 million Norwegian kroner (NOK) (increased from 6 million NOK in 2019).
- Its establishment was proposed by the Norwegian mathematician Sophus Lie when he learned that Alfred Nobel’s plans for annual prizes would not include a prize in mathematics.
- The laureates are selected by the Abel Committee, the members of which are appointed by the Norwegian Academy of Science and Letters.
Has any Indian ever won this prestigious prize?
- Srinivasa Varadhan, an Indian-American citizen won the Abel Prize in the year 2007 for his valuable contribution in “probability theory and in particular for creating a unified theory of large deviation”.
Are you an IAS Worthy Aspirant? Get a reality check with the All India Smash UPSC Scholarship Test

Get upto 100% Scholarship | 900 Registration till now | Only 100 Slots Left
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Nobel Prize
Mains level: Not Much

The Nobel Prize for Economics in 2022 was awarded to Ben S Bernanke, Douglas W Diamond and Philip H Dybvig for research on banks and financial crises.
Do you know?
- The economics prize is not one of the original five awards created in the 1895 will of industrialist and dynamite inventor Alfred Nobel.
- It was established by Sweden’s central bank and first awarded in 1969, its full and formal name being the Sveriges Riksbank Prize in Economic Sciences in Memory of Alfred Nobel.
Why was the Nobel given to these three scholars?
- The research of the three laureates has helped us understand the role of banks in the economy, particularly during financial crises.
- Their research shows why avoiding a bank collapse is very important for the economy.
Which bank did these scholars study?
- In 1656, the then-king of Sweden approved the foundation of Sweden’s first bank, the Stockholms Banco, which also became the first bank to issue banknotes in Europe.
- However, Banco over-issued notes leading to its liquidation in 1667.
- In 1668, the Swedish Nobles decided to found the Riksens Standers Bank, which was later renamed as Sveriges Riksbank in 1867.
- In 1968, on its tercentenary, the Sveriges Riksbank decided to award the economics prize in memory of Alfred Nobel.
- The award itself was the result of an ongoing crisis and conflict between the central bank and the government.
- The purpose of mentioning this history is to highlight how failures are central to banks.
- Banks have failed ever since they were created.
What does Ben Bernanke say about banking crises?
- In the 1930s, the world economy faced a serious economic contraction called the Great Depression.
- For many years, it was thought the Great Depression was due to a lack of policy stimulus.
- The economist John Maynard Keynes had argued that monetary policy was ineffective in such crises as interest rates could not go lower than zero percent, and one needed a large fiscal stimulus.
- Milton Friedman argued that central banks could create money even when interest rates were zero by buying assets, thereby increasing the money supply.
Reasons behind the crisis
- Bernanke said that while a lack of policy stimulus explains the contraction, it does not explain why the Great Depression continued for such a long time.
- The economic contraction had led to a large number of bank failures.
- His argument was that it was this large-scale failure of banks which prolonged the crisis.
- Banks were not in a position to channel loans towards productive activities, leading to the crisis becoming more severe in the US.
How is bank failure attributed to the financial crisis?
- Banks have special insights into companies, and when a bank fails, all this information is lost.
- A failed banking system takes many years to repair and the economy performs very poorly in this period.
- This explains why the Great Depression became such a prolonged crisis.
- Bernanke drew his analysis from a deep understanding of economic and monetary history.
- This prize also shows the importance of history, which is becoming rarer in economic research.
What are Diamond’s and Dybvig’s insights into banking crises?
- Bernanke explained what happens when banks fail. But Diamond and Dybvig explained why banks fail.
- In joint research, hence called the Diamond-Dybvig model, they explain that banks fail when depositors rush for their money.
- In their model, banks are seen as financial intermediaries that intermediate funds from depositors to loan seekers.
- The deposits are for shorter durations whereas loans are typically given for longer durations (technically called the maturity transformation function of banks).
- The banks are seen as entities that help savers meet investors, and by channeling loans towards good projects, banks help an economy grow.
How bank failure is related to depositors?
- Banks are prone to runs by depositors.
- In their research, they show that once there is a rumor about a bank’s weakness, it spreads like wildfire, causing a bank run, when depositors literally run for their funds to the bank.
- As banks lend most of the funds towards long-term projects, the loans cannot be recalled easily to repay the depositors.
- If the rumor is not addressed, it leads to eventual bank failure.
Is it Nobel-worthy?
- While many know this is basically how banks fail, the prize-winning duo formalized the model.
- They also presented a solution for bank failures via deposit insurance, which was also introduced before their research.
- In 1933, the US was the first country to adopt deposit insurance, followed by India in 1962.
- Both adopted deposit insurance after a significant number of banks failed in these countries.
What does the prize mean for Indian banking?
- India has been facing sporadic banking crises from 2013 where few banks failed.
- Bernanke’s research shows how once a crisis starts, it can prolong not just banking problems but also lower economic growth over time.
- Diamond-Dybvig’s research shows how the weak performance of individual banks like the Punjab and Maharashtra Urban Cooperative Bank and Yes Bank lead to runs.
- Such banks need to be bailed out by the government.
- There was also the case of ICICI bank which faced a run in 2008 based on rumours, but the run was stalled by the central bank by issuing a notification assuring the sound health of the bank.
You must know this!
- Economist and former Reserve Bank of India (RBI) chief Raghuram Rajan seemed to have missed out on the award.
- He is a leading scholar on banking and has written many research papers with this year’s awardee, Douglass Diamond.
- The Nobel committee has cited 12 of his research papers, which are a significant contribution to the field of banking.
UPSC 2023 countdown has begun! Get your personal guidance plan now! (Click here)
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Click Chemistry
Mains level: Read the attached story

Scientists Carolyn R. Bertozzi, Morten Meldal and K. Barry Sharpless won the 2022 Nobel Prize in Chemistry on for discovering reactions that let molecules snap together to create desired compounds and that offer insight into cell biology.
What the scientists worked on?
- Sharpless came up with the term ‘Click Chemistry’ and worked extensively on it,
- Meldal came up with a special chemical structure called ‘Triazole’ which has many significant applications, and
- Bertozzi took the next step of developing click reactions that could work inside living organisms — ‘bioorthogonal’ reactions take place living systems without interfering with native biochemical processes.
What is Click Chemistry?
- Chemists often try to recreate complex chemical molecules found in nature, and this has applications, among other things, in the field of medicine – how to target and block pathogens in cells.
- However, this process can be complicated and time-consuming.
- Instead of trying to wrangle reluctant carbon atoms into reacting with each other, Barry Sharpless encouraged his colleagues to start with smaller molecules that already had a complete carbon frame.
- If chemists choose simple reactions – where there is a strong intrinsic drive for the molecules to bond together – they avoid many of the side reactions, with a minimal loss of material.
Applications of click chemistry
- Meldal through his experiments came up with the useful chemical structure called triazoles, whch are stable and are found in pharmaceuticals, dyes and agricultural chemicals.
- He also found that the reaction he used could bind together numerous different molecules.
- Bertozzi, using the work of Sharpless and Meldal, came up with an efficient and innovative method to map glycans, which are carbohydrate-based polymers made by all living organisms.
UPSC 2023 countdown has begun! Get your personal guidance plan now! (Click here)
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Nobel Prize, Neanderthal, Hominins, Denisovians
Mains level: Not Much

Swedish scientist Svante Paabo won the Nobel Prize in medicine for his discoveries on human evolution that provided key insights into our immune system and what makes us unique compared with our extinct ancestors.
Svante Paabo: His work, explained
- Svante Paabo’s seminal discoveries provide the basis for exploring what makes us uniquely human.
- Hominins refer to the now-extinct species of apes that are believed to be related to modern humans, as well as modern humans themselves.
- Paabo found that gene transfer had occurred from these now extinct Hominins to Homo sapiens following the migration out of Africa around 70,000 years ago.
- This ancient flow of genes to present-day humans has physiological relevance today, for example affecting how our immune system reacts to infections.
- Paabo established an entirely new scientific discipline, called paleogenomics that focuses on studying the DNA and genetic information of extinct hominins through reconstruction.
What is the relation between evolution and biology?
- Paabo’s discoveries have established a unique resource, which is utilized extensively by the scientific community to better understand human evolution and migration.
- We now understand that archaic gene sequences from our extinct relatives influence the physiology of present-day humans.
How did Paabo establish the linkage?
- Paabo extracted DNA from bone specimens from extinct hominins, from Neanderthal remains in the Denisova caves of Germany.
- The bone contained exceptionally well-preserved DNA, which his team sequenced.
- It was found that this DNA sequence was unique when compared to all known sequences from Neanderthals and present-day humans.
- Comparisons with sequences from contemporary humans from different parts of the world showed that gene flow, or mixing of genetic information among a species, had also occurred between Denisova and Homo sapiens – the species of modern-day humans.
- This relationship was first seen in populations in Melanesia (near Australia) and other parts of South East Asia, where individuals carry up to 6% Denisova DNA.
- The Denisovan version of the gene EPAS1 confers an advantage for survival at high altitudes and is common among present-day Tibetans.
What are the challenges in carrying out such research?
- There are extreme technical challenges because with time DNA becomes chemically modified and degrades into short fragments.
- The main issue is that only trace amounts of DNA are left after thousands of years, and exposure to the natural environment leads to contamination with DNA.
Back2Basics: Neanderthal Man

- Neanderthals were humans like us, but they were a distinct species called Homo Neanderthalensis.
- Together with an Asian people known as Denisovans, Neanderthals are our closest ancient human relatives. Scientific evidence suggests our two species shared a common ancestor.
- Current evidence from both fossils and DNA suggests that Neanderthal and modern human lineages separated at least 500,000 years ago. Some genetic calibrations place their divergence at about 650,000 years ago.
- The best-known Neanderthals lived between about 130,000 and 40,000 years ago, after which all physical evidence of them vanishes.
- They evolved in Europe and Asia while modern humans – our species, Homo sapiens – were evolving in Africa.
UPSC 2023 countdown has begun! Get your personal guidance plan now! (Click here)
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Ramon Magsaysay
Mains level: Not Much

A leftist leader and former Kerala health minister has declined to receive the Ramon Magsaysay Award as the late President of the Philippines was known for his alleged brutality against the Communists.
What is the news?
- Lunched in 1958, the Ramon Magsaysay Award, widely considered to be Asia’s equivalent to the Nobel Prize, recognises outstanding leadership and communitarian contributions in Asia.
- This year’s annual list of awardees was announced last week.
- Shailaja was considered for the award for her performance as state health minister from 2016 to 2021, a period which saw Kerala fight against the Nipah virus and Covid-19.
Who was Ramon Magsaysay?
- Born on August 31, 1907 to a father who worked as a blacksmith and a mother who was a teacher, Ramon Del Fierro Magsaysay Senior was the seventh president of the Philippines, from 1953 until his death in an air crash in 1957.
- Magsaysay started out as an automobile mechanic before being drafted into the Pacific War (1941-1945), during World War II.
- The Pacific War would see the Japanese occupation of the Philippines — then a colony of the US — for nearly four years.
- The US formally recognised the Philippines as an independent nation in 1946.
His legacy
- As a guerrilla leader resisting the Japanese occupation, Magsaysay’s bravery and leadership saw his appointment as a military governor.
- In 1946, he would be elected under the Liberal Party to the Philippine House of Representatives, where he would serve two terms as a Congressman before being appointed secretary of National Defence in 1950.
- On December 30, 1953, he would be elected president from the Nationalist Party, the oldest political party in the Philippines.
Communism and Magsaysay
- Founded in 1902, the Unión Obrera Democrática is considered the first modern trade federation in the Philippines.
- The Communist Party of the Philippines or the Partido Komunista ng Pilipinas (PKP) was formed in 1930.
- The Hukbong Bayan Laban sa Hapon or the People’s Army Against the Japanese, popularly known as the Hukbalahap (Huk), was a prominent guerrilla outfit that fought the Japanese.
- As the country plunged into post-war chaos after 1946, the fortunes of the Hukbalahaps, too, changed dramatically.
How he rose to his fame?
- With the expansion of capitalism, the gap between the rich and poor widened and the farmers continued to languish.
- Huk leaders were viewed with suspicion over their declaration of commitment to communism and the demand for peasant rights.
- With the US as its close ally, the Philippine government cracked down on the Huks, who formed an alliance with the PKP to take their struggle to a parliamentary platform.
- The severe crackdown against the Huks continued until Magsaysay became the National Defence Secretary under President Elpidio Quirino.
- Magsaysay drew upon his own experience of guerrilla warfare to initiate a two-pronged system of reforms and military campaigns.
- It was under his administrative and military policies that the Huk threat was considered to be neutralised.
The Ramon Magsaysay Award
- In 1957, the Ramon Magsaysay award was set up by trustees of the Rockefeller Brothers Fund and the Philippine government to carry forward Magsaysay’s legacy of service to the people, good governance, and pragmatic idealism.
- In the six decades since 1958 — the first year the Award was given out — over 300 organisations and individuals have been recognised for their developmental endeavours crucial to Asia, and, consequently, to the world.
- The award is given out every year on August 31, on Magsaysay’s birth anniversary.
Indian winners on the list
- Prominent Indians who have won the award include Vinoba Bhave in 1958, Mother Teresa in 1962, Kamaladevi Chattopadhyay in 1966, Satyajit Ray in 1967, Mahasweta Devi in 1997.
- In recent years, Arvind Kejriwal (2006), Anshu Gupta of Goonj (2015), human rights activist Bezwada Wilson (2016), and journalist Ravish Kumar (2019) have won the award.
UPSC 2023 countdown has begun! Get your personal guidance plan now! (Click here)
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Fields Medal
Mains level: NA

Ukrainian mathematician Maryna Viazovska was named as one of four recipients of the prestigious Fields Medal, which is often described as the Nobel Prize in mathematics.
What is Fields Medal?
- The Fields Medal is awarded by the International Mathematical Union (IMU), an international non-governmental and non-profit scientific organisation.
- It is awarded every four years to one or more mathematicians under the age of 40 in recognition of “outstanding mathematical achievement for existing work and for the promise of future achievement”.
- The winners are announced at the International Congress of Mathematicians (ICM), which was supposed to be held in Russia this year, but was moved to Helsinki.
Belongings of the award
- The honour carries a physical medal of 14K gold, 63.5 mm in diameter and weighing 169 g, and with a unit price of approximately 5,500 Canadian dollars.
- There is also a cash award of CAD 15,000.
- The obverse of the medal is embossed with the head of Archimedes facing right, and some Latin quotes.
History of the Medal
- According to the IMU website, the 1924 ICM in Toronto adopted a resolution that at each conference, two gold medals would be awarded to recognise outstanding mathematical achievement.
- The Canadian mathematician Prof J C Fields, who was secretary of the 1924 Congress, later donated funds to establish the medals, which were named in his honour.
- In 1966, it was agreed that, in light of the great expansion of mathematical research, up to four medals could be awarded at each Congress.
Indian-origin winners
- Among the more than 60 mathematicians who have been awarded the Fields Medal since 1936, there are two of Indian origin.
- Akshay Venkatesh of the Institute for Advanced Study at Princeton, won in 2018, the last time the honour was announced.
- Manjul Bhargava of the Department of Mathematics at Princeton University was awarded in 2014.
UPSC 2023 countdown has begun! Get your personal guidance plan now! (Click here)
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Man Booker Price
Mains level: NA
Author Geetanjali Shree’s translated Hindi novel, Tomb of Sand, became the first Indian language book to win the International Booker Prize.
Note: Such topics hold very little relevance for CSE prelims. However, last year experience make such topics more uncertain. Still such topics hold relevance for other exams such as CAPF and state PSCs.
What is the Booker Prize?
- The Booker Prize is one of the best-known literary awards for fiction writing in English, including both novels and collections of short stories.
- It was first awarded in 1969.
- Every year a panel of judges decides the best work of the year, with the criteria being that it must be written in English and published in the UK and Ireland.
- This panel of judges is picked from among eminent cultural historians, writers, professors, and novelists, and others from related fields.
- For the Booker Prize, the winner receives £50,000.
About the book
- The 2018 novel titled ‘Ret Samadhi’ was translated by Daisy Rockwell and published as ‘Tomb of Sand’ in 2021.
- The prize is one of two literary awards given out annually by the Booker Prize Foundation, a charity whose stated aim is to “promote the art and value of literature for the public benefit”.
What about the International Booker Prize?
- The International Booker Prize began in 2005.
- A biennial prize initially, it was then awarded for a body of work available in English, including translations, with Alice Munro, Lydia Davis and Philip Roth becoming some of the early winners.
- In 2015, the rules of the International prize changed to make it an annual affair.
- The new rules stipulated that it will be awarded annually for a single book, written in another language and translated into English.
- The £50,000 prize money is divided equally between the author and translator each year.
Why is it called the ‘Booker’?
- The Booker Prize, from 1969 to 2001, was named simply after the Booker Group Limited – a British food wholesale operator that was its initial sponsor.
- The Man Group, an investment management firm based in the UK, began to sponsor the prize in 2002 and it thus came to be known as The Man Booker Prize.
- The Man Group ended their sponsorship in 2019.
- Crankstart, an American charitable foundation, has been the sponsor after that. The prize name has changed back to the ‘Booker’ since then.
Who have been some prominent winners?
- Prominent winners of the coveted prize include Margaret Atwood (‘The Testaments’), Yann Martel (‘Life of Pi’), and Julian Barnes (‘The Sense of an Ending’).
- Many Indian-origin writers have won the Booker in the past, such as Arundhati Roy (‘The God of Small Things’), Salman Rushdie (‘Midnight’s Children’), Kiran Desai (‘The Inheritance of Loss’), and Aravind Adiga (‘The White Tiger’).
- Shree is the first Indian to win an international prize.
UPSC 2023 countdown has begun! Get your personal guidance plan now! (Click here)
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Pulitzer Prize
Mains level: NA

A team of four Indian photographers have won the 2022 Pulitzer Prize for feature photography for their coverage of the Covid-19 crisis in India.
About Pulitzer Prize
- The Pulitzer is the most coveted award for journalists from across the world.
- It is announced by America’s Columbia University and bestowed on the recommendation of the Pulitzer Prize Board.
Who was Joseph Pulitzer, after whom the awards are named?
- Born to a wealthy family of Magyar-Jewish origin in Mako, Hungary, in 1847, Joseph Pulitzer had a stint in the military before he built a reputation of being a “tireless journalist”.
- In the late 1860s he joined the German-language daily newspaper Westliche Post, and by 25 he had become a publisher.
- In 1884, he was elected to the US House of Representatives from New York’s ninth district as a Democrat.
When were the Pulitzer awards instituted?
- The awards were instituted according to Pulitzer’s will, framed in 1904, where he made a provision for the establishment of the Pulitzer Prizes as an incentive to excellence.
- Pulitzer specified solely four awards in journalism, four in letters and drama, one for education, and five travelling scholarships.
- After his death in 1911, the first Pulitzer Prizes were awarded in June, 1917.
Indians who have won the Pulitzer
- A member of the Ghadar Party in America, journalist Gobind Behari Lal, was the first from India to win the Prize in 1937.
- In 2000, London-born Indian-American writer Jhumpa Lahiri won the Pulitzer Prize in Fiction for her debut short story collection Interpreter of Maladies.
- In 2003, Mumbai-born Geeta Anand was part of the team at Wall Street Journal that won a Pulitzer Prize for reporting on corporate corruption.
- In 2016, Indian-American Sanghamitra Kalita, then managing editor of Los Angeles Times, won the Pulitzer.
The list goes on to date ….
UPSC 2023 countdown has begun! Get your personal guidance plan now! (Click here)
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Abel Prize
Mains level: Not Much
The Norwegian Academy of Science and Letters has awarded the Abel prize for the year 2022 to American Mathematician Dennis Parnell Sullivan, for his contributions to topology in its broadest sense, and in particular its algebraic, geometric and dynamical aspects.
Abel Prize
- The Abel Prize is a prize awarded annually by the King of Norway to one or more outstanding mathematicians.
- It is named after Norwegian mathematician Niels Henrik Abel (1802–1829) and directly modeled after the Nobel Prizes.
- It comes with a monetary award of 7.5 million Norwegian kroner (NOK) (increased from 6 million NOK in 2019).
- Its establishment was proposed by the Norwegian mathematician Sophus Lie when he learned that Alfred Nobel’s plans for annual prizes would not include a prize in mathematics.
- The laureates are selected by the Abel Committee, the members of which are appointed by the Norwegian Academy of Science and Letters.
Has any Indian won this prestigious prize?
- R. Srinivasa Varadhan, an Indian-American citizen won the Abel Prize in the year 2007 for his valuable contribution in “probability theory and in particular for creating a unified theory of large deviation”.
UPSC 2022 countdown has begun! Get your personal guidance plan now! (Click here)
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Natural Experiments, Nobel Prize
Mains level: Not Much

The 2021 Nobel Prize in Economic Sciences was awarded to three US-based economists.
Do you know?
The Nobel Prize is officially called as Sveriges Riksbank Prize!
Who are the awardees?
- Nobel Committee awarded half the Prize to David Card for his “empirical contributions to labour economics”
- Other half to Guido Imbens and Joshua Angrist “for their methodological contributions to the analysis of causal relationships”
What makes this year’s award special?
- This is the first time the economic prize has been divided in this fashion with one half going to one awardee and other half divided across two awardees.
- In the past, prize money was divided equally between the awardees even if the prize was for different topics as is the case this time around.
- It may appear that the Nobel Prize has been given for two different contributions, but there is a common theme: “natural experiments.”
What are Natural Experiments?
- Economists are often interested in causal questions such as the impact of education on incomes, impact of COVID-19 on poverty and so on.
- They are also interested is understanding the direction of causality.
- Economists have used two kinds of experiments to study these causality and direction of causality questions: random experiments and natural experiments.
(I) Random experiments
- Under randomized experiments, the researchers allocate say medicines to a treatment group and compare the effect of the medicine with the control group which is not given the medicine.
- In 2019, the Nobel Committee gave awards to three scholars for their contribution to the field of randomized experiments.
- However, one cannot randomize experiments to study issues such as why certain people and regions are more unequal or have fewer educational opportunities and so on.
(II) Natural experiments
- In natural experiments, economists study a policy change or a historical event and try to determine the cause and effect relationship to explain these developments.
- The trio used such natural experiments to make some landmark contributions to economic development.
- Natural experiments are more difficult for two reasons. The first is to identify what will serve as a natural experiment.
- Second, in a random experiment, the researcher knows and controls the treatment and control groups which allows them to study the cause and effect of medicine.
- But in natural experiments, such clear differentiation is not possible because people choose their groups on their own and even move between the two groups.
- Despite the limitations, the researchers could use the natural setting to answer some big policy questions.
Natural experiments conducted by David Card
- One question of interest for policymakers is to understand the impact of higher minimum wages on employment.
- Earlier studies showed that increasing minimum wages leads to lower unemployment.
- Economists were also not sure of the direction of causation between minimum wages and employment.
- Say a slowdown in the economy leads to higher unemployment amid lower income groups.
- This could lead to lower income groups demanding higher minimum wages. In such a case, it is higher unemployment which leads higher minimum wages.
Contribution of Angrist and Imbens
- Angrist and Imbens showed how natural experiments can be used to identify cause and effect precisely.
- We have discussed above how natural experiments make it difficult to separate control and treatment groups. This makes it difficult to establish causal relations.
- In the 1990s, the duo developed a methodology – Local Average Treatment Effect (or LATE) – which uses a two-step process to help grapple with these problems of natural experiments.
- Say, one is interested in finding the impact of an additional year of schooling on the incomes of people.
- By using the LATE approach, they showed that effect on income of an additional year of education is around 9%.
- While it may not be possible to determine individuals in the group, one can estimate the size of the impact.
What is the importance of the award today?
- Earlier it was difficult to identify natural experiments and even if one identified them, it was difficult to generate data from these experiments.
- With increased digitalization and dissemination of archival records, it has not just become easier to identify natural experiments but also get data.
- Economists have been using natural experiments to help us understand the impact of past policies.
- As the 2020 pandemic struck, economists used the natural experiments approach extensively to analyse how previous pandemics impacted different regions and tried to draw policy lessons.
India context
- The methodology date back to the early and mid-90s and they have already had a tremendous influence on the research undertaken in several developing countries such as India.
- For instance, in India, too, it is commonly held that higher minimum wages will be counterproductive for workers.
- It is noteworthy that last year, in the wake of the Covid-induced lockdowns, several states, including UP, had summarily suspended several labour laws.
- This included the ones regulating minimum wages, arguing that such a move will boost employment.
- The main learning is that minimum wages can be increased in India without worrying about reducing employment.
UPSC 2022 countdown has begun! Get your personal guidance plan now! (Click here)
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Nobel Price, BioCatalysts
Mains level: NA

(1) Nobel Prize for Economic Sciences, 2021

The 2021 Nobel Prize in Economic Sciences has been awarded in one half to Canadian-born David Card and the other half jointly to Israeli-American Joshua D Angrist and Dutch-American Guido W Imbens.
- David Card has been awarded for his empirical contributions to labor economics. Joshua D Angrist and Guido W Imbens won the award “for their methodological contributions to the analysis of causal relationships.”
- The 2020 Nobel Prize in Economic Sciences was awarded to Paul R Milgrom and Robert B Wilson “for improvements to auction theory and inventions of new auction formats”.
Contributions
- David Card: He has analyzed how minimum wages, immigration and education impact the labor market.
- One of the significant findings of this research was that“increasing the minimum wage does not necessarily lead to fewer jobs”.
- It also led to the understanding that“people who were born in a country can benefit from new immigration, while people who immigrated at an earlier time risk being negatively affected”.
- It also illuminated the role of resources available in school in shaping the future of students in the labor market.
- Joshua Angrist and Guido Imbens: They were rewarded for their “methodological contributions” to the research tool.
- Their work demonstrated “how precise conclusions about cause and effect can be drawn from natural experiments”.
(2) Nobel Prize for Chemistry, 2021

The 2021 Nobel Prize in Chemistry was awarded to Benjamin List and David MacMillan for the development of asymmetric organocatalysis.
- Last year, the honour went to Frenchwoman Emmanuelle Charpentier and American Jennifer Doudna, for developing the gene-editing technique known as CRISPR-Cas9 – DNA snipping “scissors”.
About the Development
- They have developed a new and ingenious tool for molecule building: organocatalysis.
- Many research areas and industries are dependent on chemists’ ability to construct molecules that can form elastic and durable materials, store energy in batteries or inhibit the progression of diseases. This work requires catalysts.
- According to researchers, there were just two types of catalysts available: metals and enzymes. Catalysts are any substance that increases the rate of a reaction without itself being consumed.
- In 2000, they, independent of each other, developed a third type of catalysis. It is called asymmetric organocatalysis and builds upon small organic molecules.
- Significance:
- Its uses include research into new pharmaceuticals and it has also helped make chemistry greener.
- Both these sets of catalysts (metals and enzymes) had limitations.
- Heavier metals are expensive, difficult to mine, and toxic to humans and the environment.
- Despite the best processes, traces remained in the end product; this posed problems in situations where compounds of very high purity were required, like in the manufacture of medicines.
- Also, metals required an environment free of water and oxygen, which was difficult to ensure on an industrial scale.
- Enzymes on the other hand, work best when water is used as a medium for the chemical reaction. But that is not an environment suitable for all kinds of chemical reactions.
Organocatalysis
-
- Organic compounds are mostly naturally-occurring substances, built around a framework of carbon atoms and usually containing hydrogen, oxygen, nitrogen, sulphur, or phosphorus.
- Life-supporting chemicals like proteins, which are long chains of amino acids (carbon compounds containing nitrogen and oxygen) are organic.
- Enzymes are also proteins, and therefore, organic compounds. These are responsible for many essential biochemical reactions.
- Organocatalysts allow several steps in a production process to be performed in an unbroken sequence, considerably reducing waste in chemical manufacturing.
- Organocatalysis has developed at an astounding speed since 2000. Benjamin List and David MacMillan remain leaders in the field, and have shown that organic catalysts can be used to drive multitudes of chemical reactions.
- Using these reactions, researchers can now more efficiently construct anything from new pharmaceuticals to molecules that can capture light in solar cells.
Asymmetric Organocatalysis
-
- The process called asymmetric organocatalysis has made it much easier to produce asymmetric molecules – chemicals that exist in two versions, where one is a mirror image of the other.
- Chemists often just want one of these mirror images – particularly when producing medicines – but it has been difficult to find efficient methods for doing this.
- Some molecules with mirror versions have different properties. An example is the chemical called carvone, which has one form that smells like spearmint and a counterpart that smells like the herb, dill.
- Different versions of the same molecule might have different effects when ingested. Then it becomes important to be able to make only the mirror image of a drug that has the desired physiological effect.
(3) Nobel Prize in Physics, 2021

The 2021 Nobel Prize in Physics is awarded with one half jointly to Syukuro Manabe, Klaus Hasselmann and the other half to Giorgio Parisi “for groundbreaking contributions to our understanding of complex physical systems.”
- This is the first time climate scientists (Manabe and Hasselmann) have been awarded the Physics Nobel. Last year, the award was given for the research into black holes.
Manabe and Hasselmann
- Awarded for work in physical modelling of Earth’s climate, quantifying variability and reliably predicting global warming.
- Demonstrated how increases in the amount of carbon dioxide in the atmosphere would increase global temperatures, laying the foundations for current climate models.
Parisi
- Awarded for “the discovery of the interplay of disorder and fluctuations in physical systems from atomic to planetary scales.”
- He “built a deep physical and mathematical model” that made it possible to understand complex systems in fields such as mathematics, biology, neuroscience and machine learning.
(4) Nobel Prize for Physiology/Medicine, 2021

Recently, two United States-based scientists, David Julius and Ardem Patapoutian have been awarded the 2021 Nobel Prize for Physiology/Medicine for their discoveries of receptors for temperature and touch.
- They have focused their work on the field of somatosensation, that is the ability of specialized organs such as eyes, ears and skin to see, hear and feel.
About the Discoveries
David Julius:
- He discovered TRPV1, a heat-sensing receptor.
- His findings on the skin’s sense of temperature was based on how certain cells react to capsaicin, the molecule that makes chili peppers spicy, by simulating a false sensation of heat.
Ardem Patapoutian
- He discovered two mechanosensitive ion channels known as the Piezo channels.
- The Piezo1 is named after the Greek word for pressure, ‘píesi’.
- He is credited for finding the cellular mechanism and the underlying gene that translates a mechanical force on our skin into an electric nerve signal.
Significance of Discoveries
-
- The findings have allowed us to understand how heat, cold and mechanical force can initiate the nerve impulses that allow us to perceive and adapt to the world around us.
- This knowledge is being used to develop treatments for a wide range of disease conditions, including chronic pain.
Back To Basics: About Nobel Prizes
- The will of the Swedish scientist Alfred Nobel established the five Nobel prizes in 1895.
- The Nobel Prizes are a set of recognition given to fields of Chemistry, Literature, Peace, Physics, and Physiology or Medicine by The Nobel Foundation.
- The Nobel Foundation is a private institution established in 1900, has ultimate responsibility for fulfilling the intentions in Alfred Nobel’s will.
- The prizes in Chemistry, Literature, Peace, Physics, and Physiology or Medicine were first awarded in 1901.
- In 1968, Sveriges Riksbank established the Sveriges Riksbank Prize in Economic Sciences in Memory of Alfred Nobel.
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Nobel Price
Mains level: Climate Change Assessment

Three scientists received the Nobel Prize in Physics for work that is essential to understanding how the Earth’s climate is changing, pinpointing the effect of human behaviour on those changes and ultimately predicting the impact of global warming.
Who are the laureates?
- The winners were Syukuro Manabe, Klaus Hasselmann of Germany, and Giorgio Parisi from Italy.
- In 2015, at a UK-based climate-focused online publication sought to identify the three most influential climate change research papers ever published.
- The paper that received the most votes was one by Syukuro Manabe and Richard Wetherald way back in 1967.
- These reports for the first time, had described the impact of carbon dioxide and water vapour on global warming.
Citation for their Climate Model

- Manabe is a senior meteorologist and climatologist at Princeton University.
- In the 1960s, he led ground-breaking research into how increased levels of carbon dioxide lead to higher temperatures on the surface of the Earth.
- This laid the foundation for the development of current climate models.
- Hasselmann is a German physicist and oceanographer who greatly advanced public understanding of climate change through the creation of a model that links climate and chaotic weather systems.
- Parisi has focused on quantum field theory and complex systems.
Why it is significant feat?
- This is the first-time climate scientists have been awarded the Physics Nobel.
- The IPCC had won the Peace Nobel in 2007, an acknowledgement of its efforts in creating awareness for the fight against climate change.
- A Chemistry Nobel was also awarded to Paul Crutzen in 1995, for his work on the ozone layer, is considered the only other time someone from atmospheric sciences has won this honour.
- The recognition of Manabe and Hasselmann, therefore, is being seen as an acknowledgement of the importance that climate science holds in today’s world.
UPSC 2022 countdown has begun! Get your personal guidance plan now! (Click here)
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Nobel Price, Genes controling senses
Mains level: Read the attached story

U.S. scientists David Julius and Ardem Patapoutian have won the Nobel Medicine Prize for discoveries on receptors for temperature and touch.
Who are the Laureates?
- David Julius and Ardem Patapoutian, working independently in the United States, made a series of discoveries in the late 1990s and early 2000s.
- They figured out the touch detectors in our body and the mechanism through which they communicate with the nervous system to identify and respond to a particular touch.
What did they discover?

- They discovered the molecular sensors in the human body that are sensitive to heat, and to mechanical pressure, and make us “feel” hot or cold, or the touch of a sharp object on our skin.
- n 1997, Dr. Julius and his team published a paper in Nature detailing how capsaicin, or the chemical compound in chili peppers, causes the burning sensation.
- They created a library of DNA fragments to understand the corresponding genes and finally discovered a new capsaicin receptor and named it TRPV1.
- This discovery paved the way for the identification of many other temperature-sensing receptors.
- They identified another new receptor called TRPM8, a receptor that is activated by cold. It is specifically expressed in a subset of pain-and-temperature-sensing neurons.
- They identified a single gene PIEZO2, which when silenced made the cells insensitive to the poking. They named this new mechanosensitive ion channel Piezo1.
How do they work?
- The human ability to sense heat or cold and pressure is not very different from the working of the many detectors that we are familiar with.
- When something hot, or cold, touches the body, the heat receptors enable the passage of some specific chemicals, like calcium ions, through the membrane of nerve cells.
- It’s like a gate that opens up on a very specific request. The entry of the chemical inside the cell causes a small change in electrical voltage, which is picked up by the nervous system.
- There is a whole spectrum of receptors that are sensitive to different ranges of temperature.
- When there is more heat, more channels open up to allow the flow of ions, and the brain is able to perceive higher temperatures.
Therapeutic implications
- Breakthroughs in physiology have often resulted in an improvement in the ability to fight diseases and disorders. This one is no different.
- There are receptors that make us feel pain. If these receptors can suppress, or made less effective, the person had felt less pain.
- Chronic pain is present is a number of illnesses and disorders. Earlier, the experience of pain was a mystery.
- But as we understand these receptors more and more, it is possible that we gain the ability to regulate them in such a way that the pain is minimized.
[Note: We will compile all Nobel Prizes into a single post once all are awarded.]
UPSC 2022 countdown has begun! Get your personal guidance plan now! (Click here)
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Next-generation DNA sequencing
Mains level: Not Much
The 2020 Millennium Technology Prize has been awarded to Shankar Balasubramanian and David Klenerman, for their development of revolutionary Next-generation DNA sequencing techniques.
About Millennium Technology Prize
- The Millennium Technology Prize is one of the world’s largest technology prizes.
- It is awarded once every two years by Technology Academy Finland, an independent fund established by Finnish industry and the Finnish state in partnership.
What is next-generation DNA sequencing?
- Next-generation sequencing (NGS) is a massively parallel sequencing technology that offers ultra-high throughput, scalability, and speed.
- The technology is used to determine the order of nucleotides in entire genomes or targeted regions of DNA or RNA.
- These technologies allow for sequencing of DNA and RNA much more quickly and cheaply than the previously used sequencing.
- NGS has revolutionized the biological sciences, allowing labs to perform a wide variety of applications and study biological systems at a level never before possible.
- More than a million base pairs can be sequenced, which translates to hundreds of genes or even the whole genome of an organism.
- This is made possible by simultaneously sequencing hundreds of pieces of DNA at the same time.
What is sequencing, btw?
- DNA (or RNA, in some viruses), the genetic material of life forms, is made of four bases (A, T, G and C; with U replacing T in the case of RNA).
- A chromosome is the duplex of a long linear chain of these – and in the DNA sequence is information – the blueprint of life.
- Life famously can replicate, and DNA replicates when an enzyme, DNA polymerase, synthesises a complementary strand using an existing DNA strand as the template.
- The breakthrough idea of Balasubramanian and Klenerman was to sequence DNA (or RNA) using this process of strand synthesis.
- They cleverly modified their ATGC bases so that each shone with a different colour.
- When copied, the “coloured” copy of DNA could be deciphered from the colours alone, using miniature optical and electronic devices.
What about the cost of all this sequencing?
- When the Human Genome Project delivered the first, near-complete sequence of our genome, the cost was estimated to have been 3 billion dollars.
- As all our chromosomes together have 3 billion base pairs, it becomes an easy calculation – One dollar per sequenced base.
- By the year 2020, NGS technologies has pushed the price for sequencing to a few thousands of rupees.
Back2Basics:
What is the Human Genome Project?
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Fukuoka Prize
Mains level: Not Much
Noted journalist P. Sainath has been selected as one of the three recipients of the Fukuoka Prize for 2021.
Fukuoka Prize
- The Fukuoka Prize is given annually to distinguished people to foster and increase awareness of Asian cultures, and to create a broad framework of exchange and mutual learning among the Asian people.
- The Prize was established in 1990 by the city of Fukuoka in Japan and the Fukuoka City International Foundation.
- The Grand Prize has earlier been awarded to Muhammad Yunus from Bangladesh, historian Romila Thapar, and sarod maestro Amjad Ali Khan. Eleven Indians have received the Fukuoka Prize so far.
- 115 people from 28 countries and areas have received the Prize in the past 30 years.
Citation for the award
- In a statement issued Mr. Sainath was described as a “very deserving recipient of the Grand Prize of Fukuoka Prize”.
- The Secretariat noted his work for creating a new form of knowledge through his writings and commentaries on rural India and for “promoting civil cooperation”.
Note4Students
From UPSC perspective, the following things are important:
Prelims level: Auction Theory, Nobel Prizes
Mains level: Auction theory and its utility
This year, the Nobel Prize for Economics was awarded to Paul R Milgrom and Robert B Wilson for “improvements to auction theory and inventions of new auction formats”.
Do you remember the 2G spectrum scam, Coalgate scam etc. that rocked the nation? Can you relate this auction theory for bidding public assets to private entities?
What is Auction?
- Essentially, it is about how auctions lead to the discovery of the price of a commodity.
- Auction theory studies how auctions are designed, what rules govern them, how bidders behave and what outcomes are achieved.
- When one thinks of auctions, one typically imagines the auction of a bankrupt person’s property to pay off his creditors.
- Indeed, this is the oldest form of auction. This simple design of such an auction — the highest open bidder getting the property (or the commodity in question) — is intuitively appealing as well.
Evolving definitions of auction
- Over time, and especially over the last three decades, more and more goods and services have been brought under auction.
- The nature of these commodities differs sharply. For instance, a bankrupt person’s property is starkly different from the spectrum for radio or telecom use.
- Similarly, carbon dioxide emission credits are quite different from the spot market for buying electricity, which, in turn, is quite different from choosing which company should get the right to collect the local garbage.
- In other words, no one auction design fits all types of commodities or seller.
The Auction Theory
Three key variables need to be understood before we move to actual propositions.
(1) Rules of the auction
- Imagine participating in an auction. Your bidding behaviour is likely to differ if the rules stipulate open bids as against closed/sealed bids.
- The same applies to single bids versus multiple bids, or whether bids are made one after another or everyone bids at the same time.
(2) Commodity or service
- The second variable is the commodity or service being put up for auction. In essence, the question is how each bidder values an item.
- This is not always easy to ascertain. In terms of telecom spectrum, it might be easier to peg the right value for each bidder because most bidders are likely to put the spectrum to the same use.
- This is called the “common” value of an object.
(3) Uncertainty
- The third variable is uncertainty.
- For instance, which bidder has what information about the object, or even the value another bidder associates with the object.
The theory
- Wilson developed the theory for auctions of objects with a common value — a value which is uncertain beforehand but, in the end, is the same for everyone”.
- Wilson showed what the “winner’s curse” is in an auction and how it affects bidding.
- As shown in the illustration, it is possible to overbid — $50 when the real value is closer to $25. In doing so, one wins the auction but loses out in reality.
- Milgrom “formulated a more general theory of auctions that not only allows common values but also private values that vary from bidder to bidder”.
- He analysed the bidding strategies in a number of well-known auction formats, demonstrating that a format will give the seller higher expected revenue when bidders learn more about each other’s estimated values.
Significance of Auction theory
- Throughout history, countries have tried to allocate resources in various ways.
- Some have tried to do it through political markets, but this has often led to biased outcomes. For Ex: The rationing of essential goods worked in State-controlled economies. People who were close to the bureaucracy and the political class came out ahead of others.
- Lotteries are another way to allocate resources, but they do not ensure that scarce resources are allocated to people who value it the most.
- Auctions, for a good reason, have been the most common tool for thousands of years used by societies to allocate scarce resources.
- When potential buyers compete to purchase goods in an auction, it helps sellers discover those buyers who value the goods the most.
- Further, selling goods to the highest bidder also helps the seller maximise his or her revenues. So, both buyers and sellers benefit from auctions.
- Whether it is the auction of spectrum waves or the sale of fruits and vegetables, auctions are at the core of allocation of scarce resources in a market economy.
What are the criticisms levelled against auctions and what are the economists contribution?
1.Issue of Winner’s Curse
- The most common one is that auctions can lead buyers to overpay for resources whose value is uncertain to them.
- This criticism, popularly known as the ‘winner’s curse’, is based on a study that showed how buyers who overpaid for U.S. oil leases in the 1970s earned low returns. Dr. Wilson was the first to study this matter.
- The rational bidders may decide to underpay for resources in order to avoid the ‘winner’s curse’, and Dr. Wilson argued that sellers can get better bids for their goods if they share more information about it with potential buyers
2.Auction formats
- Economists traditionally working on auction theory believed that all auctions are the same when it comes to the revenues that they managed to bring in for sellers. The auction format, in other words, did not matter.
- This is known as the ‘revenue equivalence theorem’.
- But Dr. Milgrom showed that the auction format can actually have a huge impact on the revenues earned by sellers.
- The most famous case of an auction gone wrong for the seller was the spectrum auction in New Zealand in 1990.
- In what is called a ‘Vickrey auction’, where the winner of the auction is mandated to pay only the second-best bid, a company that bid NZ$1,00,000 eventually paid just NZ$6 and another that bid NZ$70,00,000 only paid NZ$5,000.
- In particular, Dr. Milgrom showed how Dutch auctions, in which the auctioneer lowers the price of the product until a buyer bids for it, can help sellers earn more revenues than English auctions.
- In the case of English auctions, the price rises based on higher bids submitted by competing buyers. But as soon as some of the bidders drop out of the auction as the price rises, the remaining bidders become more cautious about bidding higher prices.
Conclusion
- The contributions of Dr. Milgrom and Dr. Wilson have helped governments and private companies design their auctions better.
- This has, in turn, helped in the better allocation of scarce resources and offered more incentives for sellers to produce complex goods.
Note4Students
From UPSC perspective, the following things are important:
Prelims level: World Food Programme
Mains level: Not Much
The Nobel Peace Prize was awarded to the World Food Programme (WFP) for feeding millions of people from Yemen to North Korea, with the coronavirus pandemic seen pushing millions more into hunger.
Tap here to read more about Nobel Prizes here at:
Nobel and other Prizes
World Food Programme
- The WFP is the food-assistance branch of the United Nations and the world’s largest humanitarian organization focused on hunger and food security.
- Founded in 1961, it is headquartered in Rome and has offices in 80 countries.
- In addition to emergency food aid, WFP focuses on relief and rehabilitation, development aid, and special operations, such as making food systems more resilient against climate change and political instability.
- It is an executive member of the United Nations Development Group, which collectively aims to fulfil the 17 Sustainable Development Goals (SDG), and has prioritized achieving SDG 2 for “zero hunger” by 2030.
Note4Students
From UPSC perspective, the following things are important:
Prelims level: CRISPR, Cas9
Mains level: Gene Editing

French-American duo Emmanuelle Charpentier and Jennifer Doudna have been awarded the 2020 Nobel Prize for the chemistry of CRISPR, which allows scientists to ‘cut-paste’ inside a genetic sequence.
Try this PYQ:
Q.What is Cas9 protein that is often mentioned in news?
(a) A molecular scissors used in targeted gene editing
(b) A biosensor used in the accurate detection of pathogens in patients
(c) A gene that makes plants pest-resistant
(d) A herbicidal substance synthesized in genetically modified crops
The CRISPR technology
- The CRISPR is an acronym for Clustered Regularly Interspaced Short Palindromic Repeats, developed in the year 2012
- CRISPR has made gene editing very easy and simple, and at the same time extremely efficient.
- The technology works in a simple way — it locates the specific area in the genetic sequence which has been diagnosed to be the cause of the problem, cuts it out, and replaces it with a new and correct sequence that no longer causes the problem.
- The technology replicates a natural defence mechanism in some bacteria that use a similar method to protect itself from virus attacks.
Working of CRISPR

- An RNA molecule is programmed to locate the particular problematic sequence on the DNA strand.
- A special protein called Cas9, often described in popular literature as ‘genetic scissor’, is used to break and remove the problematic sequence.
- A DNA strand, when broken, has a natural tendency to repair itself. But the auto-repair mechanism can lead to the re-growth of a problematic sequence.
- Scientists intervene during this auto-repair process by supplying the desired sequence of genetic codes, which replaces the original sequence.
- It is like cutting a portion of a long zipper somewhere in between and replacing that portion with a fresh segment.
- Because the entire process is programmable, it has a remarkable efficiency and has already brought almost miraculous results.
Uses of CRISPR
- There are a whole lot of diseases and disorders, including some forms of cancer, that are caused by an undesired genetic mutation.
- These can all be fixed with this technology. There are vast applications elsewhere as well. Genetic sequences of disease-causing organisms can be altered to make them ineffective.
- Genes of plants can be edited to make them withstand pests, or improve their tolerance to drought or temperature.
Ethical concerns
- In November 2018, a Chinese researcher in Shenzen created an international sensation with his claim that he had altered the genes of a human embryo that eventually resulted in the birth of twin baby girls.
- This was the first documented case of a ‘designer babies’ being produced using the new gene-editing tools like CRISPR.
- What made matters worse was that the gene-editing was probably done without any regulatory permission or oversight.













































